��Ŀ����
ʵ������һƿ�����Һ����ʦ��С��ͬѧ��Ʒ����ⶨ�÷�Һ�����������������С��ͬѧ��ȡһ����С�ձ�����������Ϊ18.2g��Ȼ�������е������������Һ�������������Ϊ33.2g��֮��һö����Ϊ10.8g������������ɰֽɰȥ���⣩�����С�ձ��з�Ӧ�����������治�������ݲ������ٴγ�����������Ϊ43.9g����ش��������⣺
��1����Ӧ�в���������������� ��
��2������÷�Һ�����������������д��������̣�����������һλС������
��3���������������δ�������ԣ�2����������Ӱ���� ��ѡ�ƫ����ƫС��������Ӱ�족����ԭ���� ��
��1��0.1g ��2��32.7%. ��3��ƫС������Ҳ������һ�������ᡣ
���������������1������ϡ���ᷴӦ����������������������ȫ��Ӧ�������е����ʵ������������������������������������Ϊ��33.2g+10.8g-43.9g=0.1g�����0.1g��
��2�� ��������Ӧ�����������Ϊx��
�ձ���ϡ�����Һ������Ϊ��33.2g-18.2g=15.0g
Fe+H2SO4�TFeSO4+H2��
98 2
x 0.1g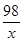 ��
��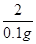
X=4.9g
�÷�Һ���������������Ϊ��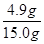 ��100%=32.7%
��100%=32.7%
�𣺷�Һ�����������������32.7%��
��3���������������δ����������������һ�������ᣬ�����������ᷴӦ������������ˮ����Ӧ����ʽΪFe2O3+3H2SO4=Fe2��SO4��3+3H2O�������������������������ƫС���Ӷ�ʹ��������������������ƫС�����ƫС������Ҳ������һ�������ᡣ
���㣺���ݻ�ѧ��Ӧ����ʽ�ļ��㣻�й��������������ļ��㣻��д��ѧ����ʽ�����ֱ���ʽ�����뷽��ʽ

С�մ�(��Ҫ�ɷ�ΪNaHCO3)�г����������Ȼ��ơ���ѧ��ȤС���ͬѧΪ�˲ⶨijƷ��С�մ���NaHCO3����������������������ʵ�飺������Ʒ�����ձ��У������������μ�ϡ���ᣬ�����ٲ�������Ϊֹ����õ��й��������±���ʾ��
| ���� | ��Ʒ | ���ĵ�ϡ���� | ��Ӧ�����Һ |
| ����(g) | 9 | 75.4 | 80 |
(1)��Ʒ�е�NaHCO3������������
(2)������Һ��NaCl������������



