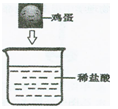
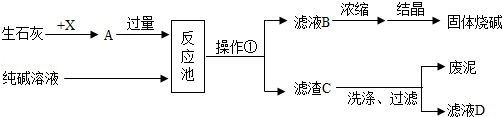
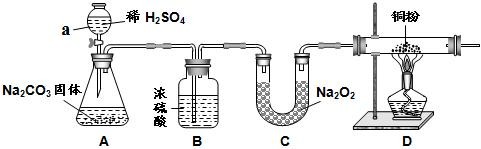
��Ŀ����
ijͬѧ��ѧϰ��Һ�������ʱ��������ҺNaCl������Na2CO3��Һ�ʼ��ԣ���ͬѧ��Ϊ�п����е�����Һ������ԣ����������Ϸ��֣�NH4��2SO4��FeCl3��Һ�����ԣ���ͬѧ����Ȥ��д�����µ�ѧϰС�ᣬ������һ����
��1������ҺpH�Ŀ���Ϊ������ļ�������ո��У����� ������ ����pH=7
��2��Ҫ�ⶨ����Һ����������� ������Ҫ֪������Һ�����ȿ����� �������������� ����
��3����ׯ��һ֦����ȫ���ʵ��ң�����NH4��2SO4��һ�ֵ����ܴٽ�ũ����ľ���Ҷ����ïʢ����ʹ��ɫʯ����Һ���� ��ɫ��
��4���̬���ʵļ�ⷽ���ǣ��� ����
��5����ͬѧ����FeCl3��Һ�����к��ɫ�������ɣ���д����ѧ����ʽ���� ��������ʾ��Cu��OH��2�������Լ���pH��5�����в��ܽ⣩
��6��ΪʲôNa2CO3��Һ�ʼ��ԣ���NH4��2SO4��FeCl3��Һ�����ԣ������鲻�Ǻ����ף��Ǹ���Ҫѧ�ģ����ϸ��к���һ��Ҫ�������
��1��pH��7��pH��7
��2�����ָʾ����pH��ֽ���ò�����պȡ����ҺͿ��pH��ֽ�ϣ�����ʾ����ɫ�����ɫ�����ռ���
��3���� ��4������ʯ�һ����ĥ���۲��Ƿ��д̼��Ե���ζ����
��5��FeCl3+3H20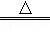 Fe��OH��3��+3HCl
Fe��OH��3��+3HCl
���������������1��pH��7����Һ�ʼ��ԣ�pH��7����Һ�����ԣ�pH=7����Һ�����ԣ�����Һ���ܳʼ��ԡ����Ի����ԣ����pH��7��pH��7��
��2�����ָʾ�����Բⶨ��Һ������ԣ��ⶨ��Һ������ʹ�õ���pH��ֽ���ⶨʱҪ�ò�����պȡ����ҺͿ��pH��ֽ�ϣ�����ʾ����ɫ�����ɫ�����ռ��ɣ�������ָʾ����pH��ֽ���ò�����պȡ����ҺͿ��pH��ֽ�ϣ�����ʾ����ɫ�����ɫ�����ռ��ɣ�
��3����NH4��2SO4��Һ�����ԣ�������Һ��ʹʯ����Һ��죬����죻
��4���̬������������ʻ�ϻ�����д̼�����ζ�İ������ʿ���ʹ������ʯ�һ����ĥ�ķ������м����̬���ʣ��������ʯ�һ����ĥ���۲��Ƿ��д̼��Ե���ζ������
��5�������Ȼ�����Һ���ɺ��ɫ�����������Ȼ�����ˮ��Ӧ���������������������Ե�ʣ���������ṩ����Ϣ����֪������pH��5��������Һ�������Լ�ܽ⣬���FeCl3+3H20 Fe��OH��3��+3HCl��
Fe��OH��3��+3HCl��
���㣺�εĻ�ѧ���ʣ���Һ�����Ȳⶨ����Һ������Բⶨ���������ʵ���������ã��̬���ʵļ��飻��д��ѧ����ʽ�����ֱ���ʽ�����뷽��ʽ��
���������⿼���˳����ε������Լ��й���Һ����ԵIJⶨ����ɴ��⣬�����������е�֪ʶ����

 ��У����ϵ�д�
��У����ϵ�д�ʵ������һƿ���ܲ������Լ�����ͼ�������ȱ�ı�ǩ��ֻʣ�¡�Na���͡�10%����������֪������ɫҺ�壬�dz��л�ѧ���õ��Լ���
��1�����������ǩ������жϣ���ƿ�Լ���������
A���� B���� C����
��2�� ��֪ ���л�ѧ�����ĺ��ƻ�������NaCl��NaOH��Na2CO3��NaHCO3��
��Na2CO3��NaHCO3��Һ���ʼ��ԡ�
�����£�20�棩ʱ���������ʵ��ܽ�ȵ���������ͼ��
| ���� | NaCl | NaOH | Na2CO3 | NaHCO3 |
| �ܽ��g | 36 | 109 | 215 | 9��6 |
�����Լ�ƿ��ע��������������10%���ұ��е��ܽ�ȵ������жϣ���ƿ�Լ��������� ��
��3�� ��pH��ֽ�ϲ�ø���Һ��pH��7����ƿ�Լ��������� ��
��4�� Ϊ��ȷ������Һ��������Һ���ֽ������½�һ����ʵ�飺
| �������� | ʵ������ | ���ۼ���ѧ����ʽ |
| ȡ�����Թ��У��μ� | �������������� | ����Һ�� ����Ӧ�Ļ�ѧ����ʽ |
��6�� Ϊ��̽��һƿ�Ȼ�����Һ������������ȡ����Һ50g������50g̼�����Һ��ǡ����ȫ��
Ӧ��������Һ����95g��������Ȼ�����Һ������������