��Ŀ����
����ϸ�Ķ�����ת����ϵ��
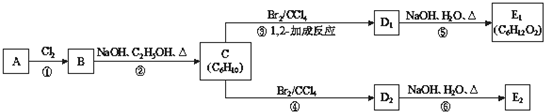
��֪����i�� �������A������ͼ��������Է�������Ϊ84��������ױ���������к���̼̼˫�����˴Ź���������ʾ�������ֻ��һ����ԭ�ӣ�
��ii�� D1��D2��Ϊͬ���칹�壬E1��E2��Ϊͬ���칹�壮
��ش�
��1��A�Ľṹ��ʽΪ ��A�����е�̼ԭ���Ƿ���ͬһƽ�棿 ����ǡ����ǡ�����
��2��д����Ӧ�ڵĻ�ѧ����ʽ����Ӧ���� ��
��3��д��E1�Ľṹ��ʽ����ϵͳ��������C������E1 ��C�����ƣ� ��
��4��д���ڼ��Ⱥ�Ũ���������£�E2������ ��Ӧ�Ļ�ѧ����ʽ�� ��
��Ӧ�Ļ�ѧ����ʽ�� ��

��֪����i�� �������A������ͼ��������Է�������Ϊ84��������ױ���������к���̼̼˫�����˴Ź���������ʾ�������ֻ��һ����ԭ�ӣ�
��ii�� D1��D2��Ϊͬ���칹�壬E1��E2��Ϊͬ���칹�壮
��ش�
��1��A�Ľṹ��ʽΪ
��2��д����Ӧ�ڵĻ�ѧ����ʽ����Ӧ����
��3��д��E1�Ľṹ��ʽ����ϵͳ��������C������E1
��4��д���ڼ��Ⱥ�Ũ���������£�E2������
 ��Ӧ�Ļ�ѧ����ʽ��
��Ӧ�Ļ�ѧ����ʽ�����㣺�л�����ƶ�
ר�⣺�л���Ļ�ѧ���ʼ��ƶ�
������ij�������A������ͼ��������Է�������Ϊ84�������ΪCxHy����x���ֵΪ
=7��������ױ��������к���̼̼˫��������AΪϩ��������x=6��y=12�����A�Ļ�ѧʽΪC6H12���˴Ź������ױ���������ֻ��һ�����͵��⣬��A�Ľṹ��ʽΪ��CH3��2C=C��CH3��2��A��������Ӧ����B��BΪ��CH3��2C��Cl��C��Cl����CH3��2��B���������ơ��Ҵ������¼��ȣ�������ȥ��Ӧ����C��CΪCH2=C��CH3��-C��CH3��=CH2��C���巢��1��2-�ӳ�����D1����D1ΪCH2BrCBr��CH3��-C��CH3��=CH2��D1����������ˮ��Һ�з���ˮ�ⷴӦ����E1��E1ΪHOCH2C��OH����CH3��-C��CH3��=CH2��D1��D2��Ϊͬ���칹�壬�ʷ�Ӧ�ܷ���1��4-�ӳɣ�D2ΪCH2BrC��CH3��=C��CH3��CH2Br��D2����������ˮ��Һ�з���ˮ�ⷴӦ����E2��E2ΪHOCH2C��CH3��=C��CH3��CH2OH���ݴ˽��
| 84 |
| 12 |
���
�⣺ij�������A������ͼ��������Է�������Ϊ84�������ΪCxHy����x���ֵΪ
=7��������ױ��������к���̼̼˫��������AΪϩ��������x=6��y=12�����A�Ļ�ѧʽΪC6H12���˴Ź������ױ���������ֻ��һ�����͵��⣬��A�Ľṹ��ʽΪ��CH3��2C=C��CH3��2��A��������Ӧ����B��BΪ��CH3��2C��Cl��C��Cl����CH3��2��B���������ơ��Ҵ������¼��ȣ�������ȥ��Ӧ����C��CΪCH2=C��CH3��-C��CH3��=CH2��C���巢��1��2-�ӳ�����D1����D1ΪCH2BrCBr��CH3��-C��CH3��=CH2��D1����������ˮ��Һ�з���ˮ�ⷴӦ����E1��E1ΪHOCH2C��OH����CH3��-C��CH3��=CH2��D1��D2��Ϊͬ���칹�壬�ʷ�Ӧ�ܷ���1��4-�ӳɣ�D2ΪCH2BrC��CH3��=C��CH3��CH2Br��D2����������ˮ��Һ�з���ˮ�ⷴӦ����E2��E2ΪHOCH2C��CH3��=C��CH3��CH2OH��
��1��������������֪��A�Ľṹ��ʽΪ����CH3��2C=C��CH3��2����CH3��2C=C��CH3��2�к���C=C˫����ƽ��ṹ��4������Cԭ�Ӵ���C=C˫����ƽ��ṹ�ڣ�����̼ԭ�Ӷ�����ͬһƽ�棬
�ʴ�Ϊ����CH3��2C=C��CH3��2���ǣ�
��2����Ӧ���ǣ�CH3��2C��Cl��C��Cl����CH3��2���������ƴ���Һ�����������·�����ȥ��Ӧ������CH2=C��CH3��-C��CH3��=CH2���÷�Ӧ�Ļ�ѧ����ʽΪ����CH3��2C��Cl��C��Cl����CH3��2+2NaOH
CH2=C��CH3��-C��CH3��=CH2+2NaCl+2H2O��
�ʴ�Ϊ��CH3��2C��Cl��C��Cl����CH3��2+2NaOH
CH2=C��CH3��-C��CH3��=CH2+2NaCl+2H2O����ȥ��Ӧ��
��3��������ķ�����֪��E1ΪHOCH2C��OH����CH3��-C��CH3��=CH2��CΪCH2=C��CH3��-C��CH3��=CH2���Ļ�ѧ������2��3-����-1��3-����ϩ��
�ʴ�Ϊ��HOCH2C��OH����CH3��-C��CH3��=CH2��2��3-����-1��3-����ϩ��
��4���ڼ��Ⱥ�Ũ���������£�HOCH2C��CH3��=C��CH3��CH2OH������ ��Ӧ�Ļ�ѧ����ʽΪ
��Ӧ�Ļ�ѧ����ʽΪ ��
��
�ʴ�Ϊ�� ��
��
| 84 |
| 12 |
��1��������������֪��A�Ľṹ��ʽΪ����CH3��2C=C��CH3��2����CH3��2C=C��CH3��2�к���C=C˫����ƽ��ṹ��4������Cԭ�Ӵ���C=C˫����ƽ��ṹ�ڣ�����̼ԭ�Ӷ�����ͬһƽ�棬
�ʴ�Ϊ����CH3��2C=C��CH3��2���ǣ�
��2����Ӧ���ǣ�CH3��2C��Cl��C��Cl����CH3��2���������ƴ���Һ�����������·�����ȥ��Ӧ������CH2=C��CH3��-C��CH3��=CH2���÷�Ӧ�Ļ�ѧ����ʽΪ����CH3��2C��Cl��C��Cl����CH3��2+2NaOH
| �� |
| �� |
�ʴ�Ϊ��CH3��2C��Cl��C��Cl����CH3��2+2NaOH
| �� |
| �� |
��3��������ķ�����֪��E1ΪHOCH2C��OH����CH3��-C��CH3��=CH2��CΪCH2=C��CH3��-C��CH3��=CH2���Ļ�ѧ������2��3-����-1��3-����ϩ��
�ʴ�Ϊ��HOCH2C��OH����CH3��-C��CH3��=CH2��2��3-����-1��3-����ϩ��
��4���ڼ��Ⱥ�Ũ���������£�HOCH2C��CH3��=C��CH3��CH2OH������
 ��Ӧ�Ļ�ѧ����ʽΪ
��Ӧ�Ļ�ѧ����ʽΪ ��
���ʴ�Ϊ��
 ��
��
���������⿼���л����ƶϣ��漰±������ϩ�����ȵ������Լ�����ʽ����⡢ͬ���칹�塢�л���ѧ��Ӧ���ͺͷ���ʽ����д�ȣ���Ŀ�ۺ��Խϴ�ע���ϩ���ļӳɷ�Ӧ���Ѷ��еȣ�

��ϰ��ϵ�д�
 ���������ν�ϵ�д�
���������ν�ϵ�д�
�����Ŀ
�ҹ������ĸ��³������У�������ɳɷ�֮һ����֪�������ͬ��Ԫ�أ���������жϴ�����ǣ�������
| A����������ɫ�Ľ��� |
| B������������������������ |
| C��������+3�۵Ļ����� |
| D�����û��������е��� |
�۲�����ģ�Ͳ�����й���Ϣ�����жϣ�����˵��������ǣ���������
| HCN | S8 | SF6���� | B12�ṹ��Ԫ | |
| �ṹģ��ʾ��ͼ |  |  |  |  |
| ��ע | / | ������CS2 | / | �۵�1873K |
| A��HCN�ĽṹʽΪH-C��N |
| B����̬��S8����ԭ�Ӿ��� |
| C��SF6���ɼ��Լ����ɵķǼ��Է��� |
| D����������ԭ�Ӿ��壬�ṹ��Ԫ�к���30��B-B�� |
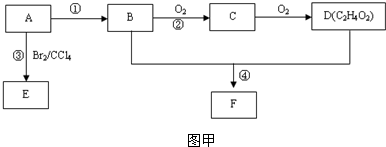




 +CH3CH2OH��
+CH3CH2OH�� +HCl��д�������ȣ�����ʽC2O2Cl2�����ӽṹʽ��
+HCl��д�������ȣ�����ʽC2O2Cl2�����ӽṹʽ�� ���������л���D��Ӧ�����ջ�ѧ����ʽ
���������л���D��Ӧ�����ջ�ѧ����ʽ �����ڼ�����--�м����--�л���A����ѡ����ȷ�м�����B��C��
�����ڼ�����--�м����--�л���A����ѡ����ȷ�м�����B��C�� C��
C��