��Ŀ����
ij��̬��A�ڱ�״���µ��ܶ�Ϊ1.25g/L�������������������һ�����ҵ�ʯ�ͻ�����չˮƽ��B��D���������г������л��D�ܸ�̼�����Ʒ�Ӧ��F����ζ������֮���ת����ϵ��ͼ����ʾ��
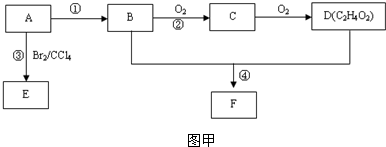
��1��A�ĽṹʽΪ ��B�й����ŵĵ���ʽΪ ��D�й����ŵ�����Ϊ ��
��2����Ӧ�ٵķ�Ӧ������ ����Ӧ�۵Ļ�ѧ����ʽΪ ��
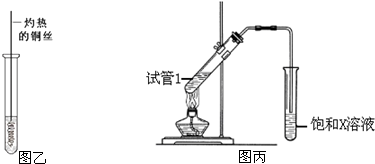
��3����Ӧ����Cu�������������½��У���ʵ��IJ����ǽ�������ͭ˿���ھƾ����ϼ��ȣ���ͭ˿��Ϊ��ɫʱ��Ѹ�ٽ�����뵽װ��B���Թ��У���ͼ����ʾ�����ظ�����2-3�Σ��÷�Ӧ�Ļ�ѧ����ʽΪ ��
��4��D��̼��������Һ��Ӧ�����ӷ���ʽΪ ��
��5��B��D��Ũ�����������ʵ�ַ�Ӧ�ܣ�ʵ��װ����ͼ����ʾ���Թ�1��װ��ҩƷ����ȣ�ͼ����X�Ļ�ѧʽΪ ���������� ���Թ�1��Ӧ�Ļ�ѧ����ʽΪ ��

��1��A�ĽṹʽΪ
��2����Ӧ�ٵķ�Ӧ������
��3����Ӧ����Cu�������������½��У���ʵ��IJ����ǽ�������ͭ˿���ھƾ����ϼ��ȣ���ͭ˿��Ϊ��ɫʱ��Ѹ�ٽ�����뵽װ��B���Թ��У���ͼ����ʾ�����ظ�����2-3�Σ��÷�Ӧ�Ļ�ѧ����ʽΪ
��4��D��̼��������Һ��Ӧ�����ӷ���ʽΪ
��5��B��D��Ũ�����������ʵ�ַ�Ӧ�ܣ�ʵ��װ����ͼ����ʾ���Թ�1��װ��ҩƷ����ȣ�ͼ����X�Ļ�ѧʽΪ

���㣺�л�����ƶ�
ר�⣺�л���Ļ�ѧ���ʼ��ƶ�
������A��һ����̬�����ڱ�״���µ��ܶ���1.25g/L��Ħ������=1.25g/L��22.4L/mol=28g/mol��������Ǻ���һ������ʯ�ͻ�����չˮƽ����AΪCH2=CH2��B��������C��C��������D��B��D���������г������л���ṹD�ķ���ʽ��֪��BΪCH3CH2OH��CΪCH3CHO��DΪCH3COOH���ʷ�Ӧ������ϩ��ˮ�����ӳɷ�Ӧ����CH3CH2OH��CH3CH2OH��CH3COOH��Ũ���������·���������Ӧ����������������FΪCH3COOCH2CH3������ϩ���巢���ӳɷ�Ӧ����1��2-�������飬�ñ��͵�̼������Һ�������������е��������Ҵ������������������ܽ�ȣ����ڷֲ㣬�ݴ˴��⣻
���
�⣺A��һ����̬�����ڱ�״���µ��ܶ���1.25g/L��Ħ������=1.25g/L��22.4L/mol=28g/mol��������Ǻ���һ������ʯ�ͻ�����չˮƽ����AΪCH2=CH2��B��������C��C��������D��B��D���������г������л���ṹD�ķ���ʽ��֪��BΪCH3CH2OH��CΪCH3CHO��DΪCH3COOH���ʷ�Ӧ������ϩ��ˮ�����ӳɷ�Ӧ����CH3CH2OH��CH3CH2OH��CH3COOH��Ũ���������·���������Ӧ����������������FΪCH3COOCH2CH3������ϩ���巢���ӳɷ�Ӧ����1��2-�������飬�ñ��͵�̼������Һ�������������е��������Ҵ������������������ܽ�ȣ����ڷֲ㣬
��1��������������֪��A�ĽṹʽΪ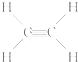 ��BΪCH3CH2OH�����й�����Ϊ�ǻ����ǻ��ĵ���ʽΪ
��BΪCH3CH2OH�����й�����Ϊ�ǻ����ǻ��ĵ���ʽΪ ��DΪCH3COOH�������Ȼ���
��DΪCH3COOH�������Ȼ���
�ʴ�Ϊ�� ��
�� ���Ȼ���
���Ȼ���
��2����Ӧ������ϩ��ˮ�����ӳɷ�Ӧ�����Ҵ�����Ӧ������ϩ���巢���ӳɷ�Ӧ����1��2-�������飬��Ӧ����ʽΪ��CH2=CH2+Br2��CH2BrCH2Br��
�ʴ�Ϊ���ӳɷ�Ӧ��CH2=CH2+Br2��CH2BrCH2Br��
��3����Cu���������������Ҵ�����������ȩ����Ӧ�Ļ�ѧ����ʽΪ2CH3CH2OH+O2
2CH3CHO+2H2O��
�ʴ�Ϊ��2CH3CH2OH+O2
2CH3CHO+2H2O��
��4��������̼��������Һ��Ӧ�����������������̼��ˮ����Ӧ�����ӷ���ʽΪ��CH3COOH+HCO3-��CH3COO-+H2O+CO2����
�ʴ�Ϊ��CH3COOH+HCO3-��CH3COO-+H2O+CO2����
��5���������������Ʊ�ʵ���У��ñ��͵�̼������Һ�������������е��������Ҵ������ܽ��Ҵ����������ᣬ���������������ķֲ㣬����XΪNa2CO3���Ʊ����������ķ�Ӧ����ʽΪ��CH3CH2OH+CH3COOH
CH3COOCH2CH3+H2O��
�ʴ�Ϊ��Na2CO3���ܽ��Ҵ����������ᣬ���������������ķֲ㣻CH3CH2OH+CH3COOH
CH3COOCH2CH3+H2O��
��1��������������֪��A�ĽṹʽΪ
 ��BΪCH3CH2OH�����й�����Ϊ�ǻ����ǻ��ĵ���ʽΪ
��BΪCH3CH2OH�����й�����Ϊ�ǻ����ǻ��ĵ���ʽΪ ��DΪCH3COOH�������Ȼ���
��DΪCH3COOH�������Ȼ����ʴ�Ϊ��
 ��
�� ���Ȼ���
���Ȼ�����2����Ӧ������ϩ��ˮ�����ӳɷ�Ӧ�����Ҵ�����Ӧ������ϩ���巢���ӳɷ�Ӧ����1��2-�������飬��Ӧ����ʽΪ��CH2=CH2+Br2��CH2BrCH2Br��
�ʴ�Ϊ���ӳɷ�Ӧ��CH2=CH2+Br2��CH2BrCH2Br��
��3����Cu���������������Ҵ�����������ȩ����Ӧ�Ļ�ѧ����ʽΪ2CH3CH2OH+O2
| Cu |
| �� |
�ʴ�Ϊ��2CH3CH2OH+O2
| Cu |
| �� |
��4��������̼��������Һ��Ӧ�����������������̼��ˮ����Ӧ�����ӷ���ʽΪ��CH3COOH+HCO3-��CH3COO-+H2O+CO2����
�ʴ�Ϊ��CH3COOH+HCO3-��CH3COO-+H2O+CO2����
��5���������������Ʊ�ʵ���У��ñ��͵�̼������Һ�������������е��������Ҵ������ܽ��Ҵ����������ᣬ���������������ķֲ㣬����XΪNa2CO3���Ʊ����������ķ�Ӧ����ʽΪ��CH3CH2OH+CH3COOH
| Ũ���� |
| �� |
�ʴ�Ϊ��Na2CO3���ܽ��Ҵ����������ᣬ���������������ķֲ㣻CH3CH2OH+CH3COOH
| Ũ���� |
| �� |
���������⿼���л����ƶϡ�����������ʵ�����Ʊ�������ϩ�봼��ȩ������֮���ת����ϵ�ǹؼ����ѶȲ���ע�����֪ʶ���������գ�

��ϰ��ϵ�д�
�����Ŀ
���б仯�У�����������������ܷ������ǣ�������
| A��SO2��S |
| B��Na2O2��O2 |
| C��Fe2+��Fe3+ |
| D��HCO3-��CO32- |
�����й����ʵ�������Ӧ�ò����Ӧ���ǣ�������
| A��SO2���������ԣ�������Ư��ֽ�� |
| B��������ˮ������Al ��OH��3���壬��������ˮ�� |
| C��FeCl3��Һ����Cu��Ӧ��������ʴ��ӡˢ��· |
| D��Zn���л�ԭ�Ժ͵����ԣ�������п�̸ɵ�صĸ������� |
���б�����ȷ���ǣ�������
| A��̼��������Һ��ˮ�ⷴӦ��HCO3-+H2O?CO32-+H3O+ | ||||
B���ö��Ե缫��ⱥ���Ȼ�þ��Һ��2Cl-+2H2O
| ||||
| C��������ĵ��뷽��ʽ��H2S=2H++S2- | ||||
| D����������þ������Һ�еμ��Ȼ�����Һ�����ֺ��ɫ����3Mg��OH��2��s��+2Fe3+��aq��?2Fe��OH��3��s��+3Mg2+��aq�� |
���з�Ӧ�����ӷ���ʽ����д������ǣ�������
| A������������Һ������������Һ��H++OH-=H2O |
| B��������ˮ���廯����Һ��Ӧ��Cl2+2Br-=2Cl-+Br2 |
| C������ˮ��2Na+2H2O=2Na++2OH-+H2�� |
| D��̼��Ƹ�ϡ���ᷴӦ��CO32-+2H+=H2O+CO2�� |
 [ѡ��5--�л���ѧ����]
[ѡ��5--�л���ѧ����]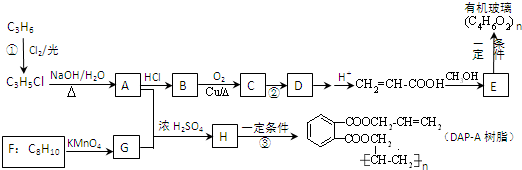
 ��ͼ�ɱ�ʾһЩ���ʻ����Ĵ�����ϵ���±��д����ѡ����ǣ�������
��ͼ�ɱ�ʾһЩ���ʻ����Ĵ�����ϵ���±��д����ѡ����ǣ�������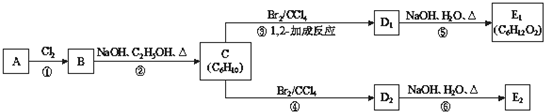
 ��Ӧ�Ļ�ѧ����ʽ��
��Ӧ�Ļ�ѧ����ʽ��