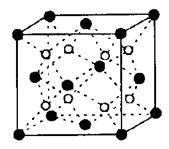
��Ŀ����
������������ͭ�Ƚ������仯�����ڹ�ҵ������Ҫ��;��
��1����̬��ԭ�ӵļ۵����Ų�ʽΪ ��
��2��CrO2Cl2��NaClO�������������������������Ȼ������Ʊ�CrO2Cl2�ķ�ӦΪ��K2Cr2O2+3CCl4 2KC+2CrO2Cl2+3COCl2����
��������Ӧʽ�зǽ���Ԫ�ص縺���ɴ�С��˳���� ����Ԫ�ط��ű�ʾ����
�ڳ���ʱCrO2Cl2��һ��������CCl4��Һ�壬���̬CrO2Cl2���� ���塣
��COCl2����������ԭ�Ӿ�����8���ӹ��ͣ�COCl2�����ЦҼ��ͦм��ĸ�����Ϊ ��
��3��NiO��FeO�ľ���ṹ�����Ȼ��Ƶľ���ṹ��ͬ������Ni2+��Fe2+�����Ӱ뾶�ֱ�Ϊ6.9��10��2nm��7.8��10��2nm�����۵㣺NiO FeO(�<���� ��=���� ��>��)��
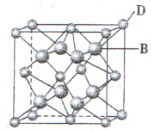
��4��CuCl��������Һ������CO���ɸ������Ȼ��ʻ���ͭ[Cu2Cl2(CO)2��2H2O]����ṹ��ͼ������˵������ȷ���� �����ţ���

��5��������ͭ��Һ�м��������ˮ��������[Cu(NH3)4]2+�����ӡ���֪NF3��NH3�Ŀռ乹�Ͷ��������Σ���NF3������Cu2+�γ������ӣ���ԭ���� ��
��1����̬��ԭ�ӵļ۵����Ų�ʽΪ ��
��2��CrO2Cl2��NaClO�������������������������Ȼ������Ʊ�CrO2Cl2�ķ�ӦΪ��K2Cr2O2+3CCl4
��������Ӧʽ�зǽ���Ԫ�ص縺���ɴ�С��˳���� ����Ԫ�ط��ű�ʾ����
�ڳ���ʱCrO2Cl2��һ��������CCl4��Һ�壬���̬CrO2Cl2���� ���塣
��COCl2����������ԭ�Ӿ�����8���ӹ��ͣ�COCl2�����ЦҼ��ͦм��ĸ�����Ϊ ��
��3��NiO��FeO�ľ���ṹ�����Ȼ��Ƶľ���ṹ��ͬ������Ni2+��Fe2+�����Ӱ뾶�ֱ�Ϊ6.9��10��2nm��7.8��10��2nm�����۵㣺NiO FeO(�<���� ��=���� ��>��)��
��4��CuCl��������Һ������CO���ɸ������Ȼ��ʻ���ͭ[Cu2Cl2(CO)2��2H2O]����ṹ��ͼ������˵������ȷ���� �����ţ���

| A���ø������д��ڻ�ѧ������ֻ�����Ӽ�����λ�� |
| B���ø�������Clԭ�ӵ��ӻ�����Ϊsp3 |
| C���ø�������ֻ��CO��H2O��Ϊ��λ�� |
| D��CO��N2�ļ۵���������ͬ����ṹΪC=O |
��13�֣���1��3d54s1��2�֣�
��2����O>Cl>C��2�֣�
�ڷ��ӣ�1�֣�
��3��1��2�֣�
��3��>��2�֣�
��4��A��C��2�֣�
��5��N��F��H����Ԫ�صĵ縺�ԣ�F>N>H����NF3�У����õ��Ӷ�ƫ��Fԭ�ӣ�ƫ��Nԭ�ӣ�ʹ��Nԭ���ϵŶԵ�������Cu2+�γ���λ���������������𰸣���2�֣�
��2����O>Cl>C��2�֣�
�ڷ��ӣ�1�֣�
��3��1��2�֣�
��3��>��2�֣�
��4��A��C��2�֣�
��5��N��F��H����Ԫ�صĵ縺�ԣ�F>N>H����NF3�У����õ��Ӷ�ƫ��Fԭ�ӣ�ƫ��Nԭ�ӣ�ʹ��Nԭ���ϵŶԵ�������Cu2+�γ���λ���������������𰸣���2�֣�
�����������1����Ϊ24��Ԫ�أ����������ԭ�������ع����֪����̬��ԭ�ӵļ۵����Ų�ʽΪ3d54s1����2����O��C��Cl��Ϊ�ǽ������ǽ����Ի�縺�ԣ�O>Cl>C����CCl4�ɷ��ӹ��ɣ�����������ԭ����֪����̬CrO2Cl2�Ƿ��Ӿ��壻���������֪��1��COCl2����1��C=O����2��C��Cl�����������ǦҼ���˫����1��Ϊ�Ҽ�����һ��Ϊ�м�����COCl2�����ЦҼ��ͦм��ĸ�����Ϊ3��1����3���������֪�������ܣ�NiO>FeO�����۵㣺NiO>FeO����4��A����ͼ��֪���ø������к��й��ۼ�������B����ԭ���Թ��ۼ�����λ����ͭ��ϣ�����ԭ�Ӳ�ȡsp3�ӻ���ʽ������C����ԭ��Ҳ�����壬����D��CO��N2��Ϊ�ȵ����壬��ȷ����5��N��F��H����Ԫ�صĵ縺�ԣ�F>N>H����NF3�У����õ��Ӷ�ƫ��Fԭ�ӣ�ƫ��Nԭ�ӣ�ʹ��Nԭ���ϵŶԵ�������Cu2+�γ���λ����

��ϰ��ϵ�д�
 ��Ӣ���㿨ϵ�д�
��Ӣ���㿨ϵ�д� Ӧ����㲦ϵ�д�
Ӧ����㲦ϵ�д� ״Ԫ����ϵ�д�
״Ԫ����ϵ�д� ͬ������ϵ�д�
ͬ������ϵ�д�
�����Ŀ
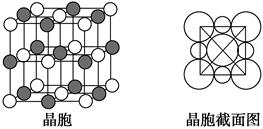
 �ڹ�����ˮ����ת��Ϊ
�ڹ�����ˮ����ת��Ϊ ��д��
��д�� �ļ۲�����Ų�ͼ____��
�ļ۲�����Ų�ͼ____�� ��
�� ����λ��Ϊ____��NH3���ӵ�����ԭ���ӻ���ʽΪ____��
����λ��Ϊ____��NH3���ӵ�����ԭ���ӻ���ʽΪ____�� �ȡ�CO��N2���ڵȵ����壬��CO������
�ȡ�CO��N2���ڵȵ����壬��CO������ ����
���� ����Ŀ��Ϊ____��д����CO��Ϊ�ȵ������һ�������ӵ����ӷ���____��
����Ŀ��Ϊ____��д����CO��Ϊ�ȵ������һ�������ӵ����ӷ���____�� O���þ�����Ni3����Ni2����������֮��Ϊ____��
O���þ�����Ni3����Ni2����������֮��Ϊ____��
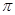 ���ĸ���Ϊ ��
���ĸ���Ϊ ��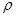 g��cm-3�������ӵ�����ΪNA�����߳�a= cm. (��
g��cm-3�������ӵ�����ΪNA�����߳�a= cm. (��