��Ŀ����
18��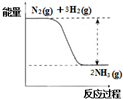 ���Ѿ����ֵ�һ�ٶ���Ԫ���У���ϡ�������⣬�ǽ���Ԫ��ֻ��ʮ���֣������������������е���ϵ��
���Ѿ����ֵ�һ�ٶ���Ԫ���У���ϡ�������⣬�ǽ���Ԫ��ֻ��ʮ���֣������������������е���ϵ����1��Ϊ�����ú�������ʣ�����������Ϊ��ȼ�����壬��Ҫ��Ӧ��̼��ˮ������Ӧ����ˮú������ѧ��Ӧ����ʽΪC��s��+H2O��g��$\frac{\underline{\;����\;}}{\;}$CO��g��+H2��g����������������H2O��
��2�����Ƕ�ֲ����������ȱ�ٵ�Ԫ�أ��ϳɰ��ķ�Ӧ����������ʳ�����Ӧ���£�N2��g��+3H2��g��$?_{����}^{���¸�ѹ}$2NH3��g����
�ٺϳɰ��ķ�Ӧ�е������仯��ͼ��ʾ���÷�Ӧ�Ƿ��ȷ�Ӧ ������ȡ����ȡ�������ԭ���Ƿ�Ӧ�ﻯѧ���������յ�������С�ڣ�����ڡ���С�ڡ��������ﻯѧ���γɷų�����������
����һ�������£���һ������N2��H2�Ļ���������ij�ܱ������У�һ��ʱ�������������˵���÷�Ӧ�ﵽƽ��״̬����D������ţ���
A��������N2��H2��NH3���� B��N2��NH3Ũ�����
C�� N2��H2��NH3�����ʵ���֮��Ϊ1��3��2 D�������е�ѹǿ����ʱ��仯
��3����ҵ���õ�ⱥ��ʳ��ˮ�ķ�����ȡ�������ռ���������仯ѧ��Ӧ����ʽΪ2NaCl+2H2O$\frac{\underline{\;���\;}}{\;}$2NaOH+H2��+Cl2�������������ռ�ɽ�һ����Ӧ���Ƶ�����Һ����Ӧ�����ӷ���ʽΪCl2+2OH-=Cl-+ClO-+H2O��
���� ��1��C��ˮ��Ӧ����CO��������ˮ��HԪ�صĻ��ϼ۽��ͣ�
��2������ͼ��֪����Ӧ�������������������������������ѻ�ѧ���������������ɻ�ѧ���ͷ�������
�ڽ��ƽ��ʱ���淴Ӧ������ȡ�����ֵ�Ũ�Ȳ���ȼ���������һЩ������������
��3����ⱥ��ʳ��ˮ�ķ�����ȡ�������ռ���������������ռӦ����NaCl��NaClO��ˮ��
��� �⣺��1��C��ˮ��Ӧ����CO����������ӦΪC��s��+H2O��g��$\frac{\underline{\;����\;}}{\;}$CO ��g��+H2��g����ˮ��HԪ�صĻ��ϼ۽��ͣ���������ΪH2O��
�ʴ�Ϊ��C��s��+H2O��g��$\frac{\underline{\;����\;}}{\;}$CO ��g��+H2��g����H2O��
��2������ͼ��֪����Ӧ������������������������������÷�ӦΪ���ȷ�Ӧ�����ѻ�ѧ���������������ɻ�ѧ���ͷ���������H=���ѻ�ѧ����������-���ɻ�ѧ���ͷ�������0����֪���ѻ�ѧ����������С�����ɻ�ѧ���ͷ�������
�ʴ�Ϊ�����ȣ�С�ڣ�
��A����ӦΪ���淴Ӧ��������N2��H2��NH3���治���ж�ƽ��״̬����A����
B��N2��NH3Ũ�����ȡ����ʼŨ����ת���ʣ������ж�ƽ��״̬����B����
C�� N2��H2��NH3�����ʵ���֮��Ϊ1��3��2��ȡ����ʼŨ����ת���ʣ������ж�ƽ��״̬����C����
D���÷�ӦΪ��Ӧǰ���������ʵ������ȵķ�Ӧ���������е�ѹǿ����ʱ��仯����֪��Ӧ�ﵽƽ��״̬����D��ȷ��
�ʴ�Ϊ��D��
��3����ⱥ��ʳ��ˮ�ķ�����ȡ�������ռ����������ӦΪ2NaCl+2H2O$\frac{\underline{\;���\;}}{\;}$2NaOH+H2��+Cl2�������������ռӦ������Һ�����ӷ���ʽΪCl2+2OH-=Cl-+ClO-+H2O���ʴ�Ϊ��2NaCl+2H2O$\frac{\underline{\;���\;}}{\;}$2NaOH+H2��+Cl2����Cl2+2OH-=Cl-+ClO-+H2O��
���� ���⿼����ۺϣ��漰������ԭ��Ӧ����ѧ��Ӧ�������仯����ѧƽ��״̬���ж��ȣ�ע�ظ�Ƶ����Ŀ��飬�����ڻ���֪ʶ�ͻ�����������������Ŀ�ѶȲ���

 �Ķ��쳵ϵ�д�
�Ķ��쳵ϵ�д�| A�� | CH3OH��CH3OCH3 | B�� | HCOOH��C17H33COOH | ||
| C�� | CH3CH2CH2Br��CH3CHBrCH3 | D�� |  �� ��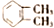 |
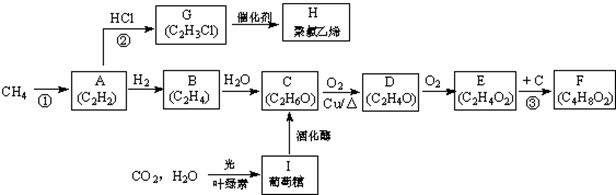
��1����Ӧ�ڵ�����Ϊ��ȥ��Ӧ��E�Ĺ����ŵ������Ȼ�
��2��д���ṹ��ʽ��DΪCH3CHO��HΪ
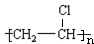
��3����E����F�Ļ�ѧ����ʽΪCH3COOH+C2H5OH$��_{��}^{Ũ����}$CH3COOC2H5+H2O����I����C�Ļ�ѧ����ʽΪC6H12O6�������ǣ�$\stackrel{�ƻ�ø}{��}$2C2H5OH+2CO2
��4����֪���Ͽ�1mol���ۼ����յ��������γ�1mol���ۼ��ͷŵ������������±�
| ���ۼ� | H-H | C-H | C��C |
| �����仯/kJ•mol-1 | 436 | 413 | 812 |
| A�� | ԭ�Ӱ뾶��O��N��C | B�� | ���ȶ��ԣ�H2O��HF��H2S | ||
| C�� | ���ԣ�KOH��NaOH��Mg��OH��2 | D�� | ���ԣ�H3PO4��H2SO4��HClO4 |
| A�� | ��Ȼ������Ҫ�ɷ��Ǽ��� | |
| B�� | ú�����л����������ɵĸ��ӵĻ���� | |
| C�� | ��C18�������������;��ѻ����Եõ����� | |
| D�� | ú�к��б��ͼױ����������ȸ�������ķ��������Ƿ������ |
| ѡ�� | ��ʵ | ���� |
| A | ��ȫȼ�ղ�����CO2��H2O������ | һ��ֻ��C��HԪ�� |
| B | ������֯Ʒ����ë���͵������� | ���ս���ë��ζ������ë�� |
| C | �ڵ�����Һ�м������۵ļӵ�ʳ�β�������ɫ | ˵������һ�������� |
| D | ���������л����Է���������ͬ���ṹ��ͬ | ����һ����ͬ���칹�� |
| A�� | A | B�� | B | C�� | C | D�� | D |
| A�� | �ﵽ��ѧƽ��ʱ������Ӧ���淴Ӧ�����ʶ�Ϊ�� | |
| B�� | �ﵽ��ѧƽ��ʱ��N2��H2��NH3�����ʵ���Ũ�Ȳ��ٱ仯 | |
| C�� | �ﵽ��ѧƽ��ʱ��N2����ȫת��ΪNH3 | |
| D�� | �ﵽ��ѧƽ��ʱ��N2��H2��NH3�����ʵ���Ũ��һ����� |
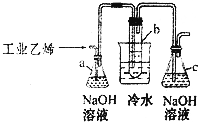 CH2Br-CH2Br�������Ϳ����������Ӽ�������������ɫҺ�壬�¶�2.18g•cm-3���е�131.4�棬�۵�9.79�棬������ˮ�������ڴ����ѡ���ͪ���л��ܼ����Թ�b��װ��Һ�壨���渲������ˮ������ҵ����ϩ�к�����H2S���壮���ù�ҵ��ϩͨ������װ�ÿ��Ʊ�����CH2B-CH2Br
CH2Br-CH2Br�������Ϳ����������Ӽ�������������ɫҺ�壬�¶�2.18g•cm-3���е�131.4�棬�۵�9.79�棬������ˮ�������ڴ����ѡ���ͪ���л��ܼ����Թ�b��װ��Һ�壨���渲������ˮ������ҵ����ϩ�к�����H2S���壮���ù�ҵ��ϩͨ������װ�ÿ��Ʊ�����CH2B-CH2Br