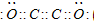��Ŀ����
����A��B��C��D��E���ֿ�����ǿ����ʣ�������ˮ�пɵ�������������ӣ��������Ӳ��ظ�����
��֪����A��B����Һ�ʼ��ԣ�C��D��E��Һ�����ԣ�
��A��Һ��E��Һ��Ӧ�����������г���������A��Һ��C��Һ��Ӧֻ�����������
��D��Һ������������Һ��Ӧ���ܲ���������Cֻ����D��Ӧ����������
������������������������
�Իش��������⣺
��1��д��A��Һ��E��Һ��Ӧ�Ļ�ѧ����ʽ��
��2��д��E��Һ�������B��Һ��Ӧ�����ӷ���ʽ��
��3����֪��NaOH��aq��+HNO3��aq��=NaNO3��aq��+H2O��l����H=-akJ?mol-1����д��B��C��ϡ��Һ��Ӧ���Ȼ�ѧ����ʽ��
| ������ | H+��Na+��Al3+��Ag+��Ba2+ |
| ������ | OH-��Cl-��CO32-��NO3-��SO42- |
��A��Һ��E��Һ��Ӧ�����������г���������A��Һ��C��Һ��Ӧֻ�����������
��D��Һ������������Һ��Ӧ���ܲ���������Cֻ����D��Ӧ����������
������������������������
�Իش��������⣺
��1��д��A��Һ��E��Һ��Ӧ�Ļ�ѧ����ʽ��
3Na2CO3+Al2��SO4��3+3H2O=2Al��OH��3��+3CO2��+3Na2SO4
3Na2CO3+Al2��SO4��3+3H2O=2Al��OH��3��+3CO2��+3Na2SO4
����2��д��E��Һ�������B��Һ��Ӧ�����ӷ���ʽ��
2A13++3SO42-+3Ba2++8OH-=2A1O2-+3BaSO4��+4H2O
2A13++3SO42-+3Ba2++8OH-=2A1O2-+3BaSO4��+4H2O
����3����֪��NaOH��aq��+HNO3��aq��=NaNO3��aq��+H2O��l����H=-akJ?mol-1����д��B��C��ϡ��Һ��Ӧ���Ȼ�ѧ����ʽ��
OH-��aq��+H+��aq��=H2O��1����H=-akJ/mol��1/2Ba��OH��2��aq��+HC1��aq��=1/2BaC12��aq��+H2O��1����H=-akJ/mol
��Ba��OH��2��aq��+2HC1��aq��=BaC12��aq��+2H2O��1����H=-2akJ/mol
��Ba��OH��2��aq��+2HC1��aq��=BaC12��aq��+2H2O��1����H=-2akJ/mol
OH-��aq��+H+��aq��=H2O��1����H=-akJ/mol��1/2Ba��OH��2��aq��+HC1��aq��=1/2BaC12��aq��+H2O��1����H=-akJ/mol
��Ba��OH��2��aq��+2HC1��aq��=BaC12��aq��+2H2O��1����H=-2akJ/mol
����Ba��OH��2��aq��+2HC1��aq��=BaC12��aq��+2H2O��1����H=-2akJ/mol
��������A��B����Һ�ʼ��ԣ�������ӵĹ����֪��ӦΪBa��OH��2��Na2CO3�е�һ�֣�C��D��E��Һ�����ԣ�ӦΪAgNO3����������HCl�е�һ�֣�
��A��Һ��E��Һ��Ӧ�����������г���������A��Һ��C��Һ��Ӧֻ�������������AΪNa2CO3��BΪBa��OH��2��EΪAl2��SO4��3��CΪHCl��
��D��Һ������������Һ��Ӧ���ܲ���������Cֻ����D��Ӧ������������DΪAgNO3��Ȼ�������ʵ����ʼ���ѧ���������
��A��Һ��E��Һ��Ӧ�����������г���������A��Һ��C��Һ��Ӧֻ�������������AΪNa2CO3��BΪBa��OH��2��EΪAl2��SO4��3��CΪHCl��
��D��Һ������������Һ��Ӧ���ܲ���������Cֻ����D��Ӧ������������DΪAgNO3��Ȼ�������ʵ����ʼ���ѧ���������
����⣺��A��B����Һ�ʼ��ԣ�������ӵĹ����֪��ӦΪBa��OH��2��Na2CO3�е�һ�֣�C��D��E��Һ�����ԣ�ӦΪAgNO3����������HCl�е�һ�֣�
��A��Һ��E��Һ��Ӧ�����������г���������A��Һ��C��Һ��Ӧֻ�������������AΪNa2CO3��BΪBa��OH��2��EΪAl2��SO4��3��CΪHCl��
��D��Һ������������Һ��Ӧ���ܲ���������Cֻ����D��Ӧ������������DΪAgNO3��
��1��A��Һ��E��Һ��Ӧ�Ļ�ѧ����ʽΪ3Na2CO3+Al2��SO4��3+3H2O=2Al��OH��3��+3CO2��+3Na2SO4��
�ʴ�Ϊ��3Na2CO3+Al2��SO4��3+3H2O=2Al��OH��3��+3CO2��+3Na2SO4��
��2��E��Һ�������B��Һ��Ӧ�����ӷ���ʽΪ2A13++3SO42-+3Ba2++8OH-=2A1O2-+3BaSO4��+4H2O��
�ʴ�Ϊ��2A13++3SO42-+3Ba2++8OH-=2A1O2-+3BaSO4��+4H2O��
��3��B��C��ϡ��Һ��Ӧ���Ȼ�ѧ����ʽΪOH-��aq��+H+��aq��=H2O��1����H=-a kJ/mol��1/2Ba��OH��2��aq��+HC1��aq��=1/2BaC12��aq��+H2O��1����H=-a kJ/mol
��Ba��OH��2��aq��+2HC1��aq��=BaC12��aq��+2H2O��1����H=-2a kJ/mol��
�ʴ�Ϊ��OH-��aq��+H+��aq��=H2O��1����H=-a kJ/mol��1/2Ba��OH��2��aq��+HC1��aq��=1/2BaC12��aq��+H2O��1����H=-a kJ/mol
��Ba��OH��2��aq��+2HC1��aq��=BaC12��aq��+2H2O��1����H=-2a kJ/mol��
��A��Һ��E��Һ��Ӧ�����������г���������A��Һ��C��Һ��Ӧֻ�������������AΪNa2CO3��BΪBa��OH��2��EΪAl2��SO4��3��CΪHCl��
��D��Һ������������Һ��Ӧ���ܲ���������Cֻ����D��Ӧ������������DΪAgNO3��
��1��A��Һ��E��Һ��Ӧ�Ļ�ѧ����ʽΪ3Na2CO3+Al2��SO4��3+3H2O=2Al��OH��3��+3CO2��+3Na2SO4��
�ʴ�Ϊ��3Na2CO3+Al2��SO4��3+3H2O=2Al��OH��3��+3CO2��+3Na2SO4��
��2��E��Һ�������B��Һ��Ӧ�����ӷ���ʽΪ2A13++3SO42-+3Ba2++8OH-=2A1O2-+3BaSO4��+4H2O��
�ʴ�Ϊ��2A13++3SO42-+3Ba2++8OH-=2A1O2-+3BaSO4��+4H2O��
��3��B��C��ϡ��Һ��Ӧ���Ȼ�ѧ����ʽΪOH-��aq��+H+��aq��=H2O��1����H=-a kJ/mol��1/2Ba��OH��2��aq��+HC1��aq��=1/2BaC12��aq��+H2O��1����H=-a kJ/mol
��Ba��OH��2��aq��+2HC1��aq��=BaC12��aq��+2H2O��1����H=-2a kJ/mol��
�ʴ�Ϊ��OH-��aq��+H+��aq��=H2O��1����H=-a kJ/mol��1/2Ba��OH��2��aq��+HC1��aq��=1/2BaC12��aq��+H2O��1����H=-a kJ/mol
��Ba��OH��2��aq��+2HC1��aq��=BaC12��aq��+2H2O��1����H=-2a kJ/mol��
���������⿼�����ӵĹ��漰���ʵ��ƶϣ���ȷ��Ϣ������֮��ķ�ӦΪ���Ĺؼ�����Ϥ����֮��ķ�Ӧ���ɽ����Ŀ�Ѷ��еȣ�

��ϰ��ϵ�д�
�����Ŀ
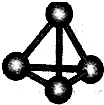 ����A��B��C��D��E��F���ֶ�����Ԫ�أ����ǵ�ԭ��������������D��E���⻯����ӹ��Ͷ���V�ͣ�A��B������������֮����C��������������ȣ�A�ֱܷ���B��C��D�γɵ���������ȵķ��ӣ���A��D���γɵĻ���������¾�ΪҺ̬��
����A��B��C��D��E��F���ֶ�����Ԫ�أ����ǵ�ԭ��������������D��E���⻯����ӹ��Ͷ���V�ͣ�A��B������������֮����C��������������ȣ�A�ֱܷ���B��C��D�γɵ���������ȵķ��ӣ���A��D���γɵĻ���������¾�ΪҺ̬��