��Ŀ����
��Դ����������������ٵ��ش���⣬�ձ����������ĺ�й©�¹����������ǶԺ���Դ�Ŀֻš����״���δ����Ҫ����ɫ��Դ֮һ��
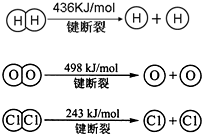
��1����֪����25�桢101kPa�£�1g�״�ȼ������CO2��Һ̬ˮʱ����22��68KJ����д���״�ȼ�յ��Ȼ�ѧ����ʽ_____________��
��2��Ŀǰ�п�ѧ����һ������������ˮú����CO��H2���ϳɼ״���

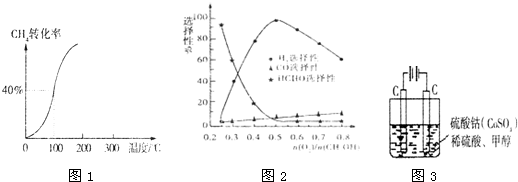
�״������ʵ����뷴Ӧ�¶ȵĹ�ϵ��ͼ��ʾ��
��1����֪����25�桢101kPa�£�1g�״�ȼ������CO2��Һ̬ˮʱ����22��68KJ����д���״�ȼ�յ��Ȼ�ѧ����ʽ_____________��
��2��Ŀǰ�п�ѧ����һ������������ˮú����CO��H2���ϳɼ״���

�״������ʵ����뷴Ӧ�¶ȵĹ�ϵ��ͼ��ʾ��
�ٺϳɼ״���Ӧ�ġ�H__________0����������� ������������
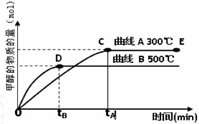
�������������䣬��E����ݻ�ѹ����ԭ����1��2������Ӧ���ʼӿ죬�淴Ӧ����___________ �� ����ӿ족�����������������䡱��������ƽ��ʱ��CH3OH����c��CO��____________��������� ����С�����䡱��
��230��ʱ��ƽ�ⳣ��K��1���������������䣬���¶����ߵ�450��ʱ���ﵽƽ��ʱ��K____________L���������������
��3�������йؼ״���˵����ȷ����____________
A���״���ʹ�����ʱ��� B���״��ܷ�����ȥ��Ӧ C���״����ܱ������� D���״����Ʒ�Ӧ��ˮ���Ʒ�Ӧ����
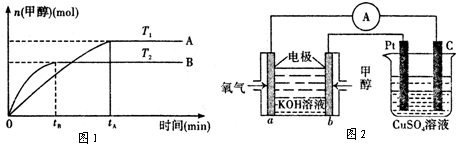
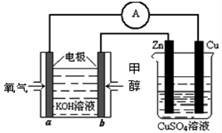
��4�����ü״�ȼ�ϵ�������ͼ��ʾ��װ�ã� д��a���ĵ缫��Ӧʽ_____________��
�������������䣬��E����ݻ�ѹ����ԭ����1��2������Ӧ���ʼӿ죬�淴Ӧ����___________ �� ����ӿ족�����������������䡱��������ƽ��ʱ��CH3OH����c��CO��____________��������� ����С�����䡱��
��230��ʱ��ƽ�ⳣ��K��1���������������䣬���¶����ߵ�450��ʱ���ﵽƽ��ʱ��K____________L���������������
��3�������йؼ״���˵����ȷ����____________
A���״���ʹ�����ʱ��� B���״��ܷ�����ȥ��Ӧ C���״����ܱ������� D���״����Ʒ�Ӧ��ˮ���Ʒ�Ӧ����
��4�����ü״�ȼ�ϵ�������ͼ��ʾ��װ�ã� д��a���ĵ缫��Ӧʽ_____________��
��1��CH3OH��l��+3/2O2��g�� CO2��g��+2H2O��l������H=��725��8 kJ��mol-1
CO2��g��+2H2O��l������H=��725��8 kJ��mol-1
��2����< �� �� �ӿ� �� ���� �� �� <
��3��A
��4��O2+4e- +2H2O==4OH-
 CO2��g��+2H2O��l������H=��725��8 kJ��mol-1
CO2��g��+2H2O��l������H=��725��8 kJ��mol-1 ��2����< �� �� �ӿ� �� ���� �� �� <
��3��A
��4��O2+4e- +2H2O==4OH-

��ϰ��ϵ�д�
 ��У����ϵ�д�
��У����ϵ�д�
�����Ŀ

 һ�������£������Ϊ3L���ܱ������У�һ����̼��������Ӧ���ɼ״�������ΪCu2O/ZnO����CO��g��+2H2��g��?CH3OH��g��
һ�������£������Ϊ3L���ܱ������У�һ����̼��������Ӧ���ɼ״�������ΪCu2O/ZnO����CO��g��+2H2��g��?CH3OH��g��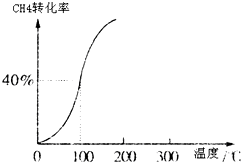 �ϳɰ���ũҵ���������������������Ҫ���壮
�ϳɰ���ũҵ���������������������Ҫ���壮