��Ŀ����
(9��) ��Ũ����ȡ������Ϊ50 mL��A��B����KOH��Һ�У��ֱ�ͨ��һ������SO2�������ϡ�͵�100 mL��
��1����KOH��Һ��ͨ��һ������SO2�������Һ�����ʵ���ɿ����ǣ�
�� �� �� ��
�� �� �� ��
��2����ϡ�ͺ����Һ����μ���0.1 mol/L�����ᣬ����SO2�����(��״��)����������������ϵ����ͼ��ʾ��
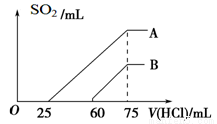
��A���߱�����ԭ��Һͨ��SO2������������������ᷴӦ����SO2����������____mL(��״��)��
��B���߱�����ԭ��Һͨ��SO2�����������Һ�����ʵĻ�ѧʽΪ_________________��
��ԭKOH��Һ�����ʵ���Ũ��Ϊ________ ____��
��9�֣�
��1���� KOH��K2SO3 �� K2SO3
�� K2SO3��KHSO3 �� KHSO3
��2���� 112 �� KOH��K2SO3
��0.150 mol/L
��������
�����������1����������������������Һ��Ӧʱ����������������������������ǡ�÷�Ӧ������������أ������������������������ǡ����ȫ��Ӧ����������������أ����Զ�������������������Һ��Ӧ�õ�����Һ�����ʿ��������ֿ��ܣ��� KOH��K2SO3 ����K2SO3���� K2SO3��KHSO3�� ��KHSO3��
��2������Ϊ����������������ᷴӦ�����������������������������������������������ɶ���������������������ȡ�A���߿�ʼ������������������������ĵ�������������������������ĵ�����������˵������Һ�е�����ΪK2SO3��KHSO3����������K2SO3��Ӧ����KHSO3����������25mL������������������ӷ�Ӧ���ɶ���������������50mL��������Һ��K2SO3��KHSO3�����ʵ�����ȣ����յõ���ҺΪ�Ȼ�����Һ����K2SO3��KHSO3�����ʵ�������x����3x=0.075L��0.1mol/L�����x=0.0025mol�����Բ�����������������2��0.0025mol��22.4L/mol=0.0112L=112mL��
�ڸ���ͼ���е����ݿ�֪�������������ĵ���������С��������������ĵ�����������˵����Һ�е�����ΪKOH��K2SO3 ��
�۸���A���ߵĽ����֪K2SO3��KHSO3�����ʵ�������0.0025mol����KԪ�ص����ʵ�����3��0.0025mol=0.0075mol������ԭKOH��Һ�����ʵ���Ũ��Ϊ0.0075mol/0.05L=0.15mol/L��
���㣺�����������������������Һ��Ӧ���жϣ�����ķ�������ͼ��ķ����жϼ�����

 ��������һ���þ�ϵ�д�
��������һ���þ�ϵ�д� Сѧ��10����Ӧ����ϵ�д�
Сѧ��10����Ӧ����ϵ�д�



