��Ŀ����
ijͬѧ�����һ����ʵ�飨��ͼ��ʾ�����Բⶨþ���Ͻ������������������ɹ�ѡ�õ��Լ��У�ŨH2SO4��Ũ���ᡢ6mol?L-1NaOH��Һ������д���пհף�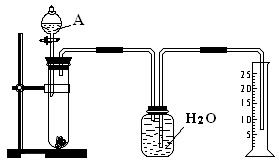
�й�ʵ������������Թ��з������������Ͻ���Ʒ���ڴӷ�Һ©�����Թ��м��������A��Һ���ۼ��װ�õ������ԣ��ܲⶨ�ռ�����ˮ�������
��1����ȷ�IJ���˳��Ϊ______��
��2��A��Һ��ѡ��______��д��ʵ���з�����Ӧ�Ļ�ѧ����ʽ��______��
��3����ʵ���в����Ʒ������ΪWg�����������ΪaL����״����������Ʒ��Al����������Ϊ______��

�й�ʵ������������Թ��з������������Ͻ���Ʒ���ڴӷ�Һ©�����Թ��м��������A��Һ���ۼ��װ�õ������ԣ��ܲⶨ�ռ�����ˮ�������
��1����ȷ�IJ���˳��Ϊ______��
��2��A��Һ��ѡ��______��д��ʵ���з�����Ӧ�Ļ�ѧ����ʽ��______��
��3����ʵ���в����Ʒ������ΪWg�����������ΪaL����״����������Ʒ��Al����������Ϊ______��
����ˮ���ռ����壬ʵ����ͨ���ⶨ������������������ݼ���ȷ���Ͻ���Al������������
��1���ռ���������������ҩƷǰ�ȼ���װ�õ������ԣ�Ȼ����Ƚ����Ͻ��ټ�������������Һ���ⶨ�ռ�����ˮ�����ȷ���������������������ȷ�IJ���˳��Ϊ�ۡ��١��ڡ��ܣ�
�ʴ�Ϊ���ۡ��١��ڡ��ܣ�
��2������ˮ���ռ����壬ʵ����ͨ���ⶨ�����������������A���Լ�ΪNaOH��Һ������������Һ������Ӧ����ƫ����������������Ӧ���ӷ���ʽΪ��2Al+2NaOH+2H2O�T2NaAlO2+3H2����
�ʴ�Ϊ��NaOH��Һ��2Al+2NaOH+2H2O�T2NaAlO2+3H2����
��3��ʵ���в����Ʒ������ΪWg�����������ΪaL����״��������
2Al+2NaOH+2H2O�T2NaAlO2+3H2����
54g 3��22.4L
m��Al�� aL
����m��Al��=54g��
=
g��
�ʺϽ���Al����������=
��100%=
��100%��
�ʴ�Ϊ��
��100%��
��1���ռ���������������ҩƷǰ�ȼ���װ�õ������ԣ�Ȼ����Ƚ����Ͻ��ټ�������������Һ���ⶨ�ռ�����ˮ�����ȷ���������������������ȷ�IJ���˳��Ϊ�ۡ��١��ڡ��ܣ�
�ʴ�Ϊ���ۡ��١��ڡ��ܣ�
��2������ˮ���ռ����壬ʵ����ͨ���ⶨ�����������������A���Լ�ΪNaOH��Һ������������Һ������Ӧ����ƫ����������������Ӧ���ӷ���ʽΪ��2Al+2NaOH+2H2O�T2NaAlO2+3H2����
�ʴ�Ϊ��NaOH��Һ��2Al+2NaOH+2H2O�T2NaAlO2+3H2����
��3��ʵ���в����Ʒ������ΪWg�����������ΪaL����״��������
2Al+2NaOH+2H2O�T2NaAlO2+3H2����
54g 3��22.4L
m��Al�� aL
����m��Al��=54g��
| aL |
| 3��22.4L |
| 18a |
| 22.4 |
�ʺϽ���Al����������=
| ||
| wg |
| 18a |
| 22.4w |
�ʴ�Ϊ��
| 18a |
| 22.4w |

��ϰ��ϵ�д�
 ��ʦָ����ĩ��̾�ϵ�д�
��ʦָ����ĩ��̾�ϵ�д� �����ܿ����ϵ�д�
�����ܿ����ϵ�д�
�����Ŀ