��Ŀ����
��12�֣�a��b��c��dΪԭ��������������Ķ�����Ԫ�أ�b��c��dͬ���ڣ���a��b��c��d����Ԫ���γɵij�����ʽ��A����ͼ��ʾ��ת����ϵ��ͼ��ÿ����ĸ��ʾһ�ֵ��ʻ����
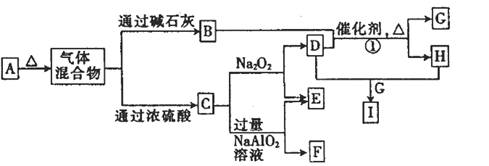
��1������C�ĽṹʽΪ________________��
��2��д���������ʵĻ�ѧʽ��F________��I________��
��3��д����Ӧ�ٵĻ�ѧ����ʽ: ______________________��
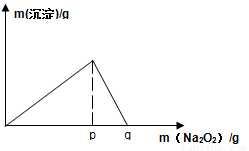
��4����F��һ���������ϣ�����ǡ�÷�Ӧ�õ���ҺM����M�м���Na2O2ʱ������Na2O2���������������������������ͼ��ʾ��ϵ����p��ʱ����������ų���������ʵ���֮��Ϊ ���ӿ�ʼ��Na2O2������q������������У��ܷ�Ӧ�����ӷ���ʽΪ ��
��4����A��E��ɵĹ�������X g ����ˮ�����Һ����������������I��ϡ��Һ����ü���I��Һ�����������C���������״�������±���ʾ����I��Һ�����ʵ���Ũ��Ϊ_____��
|
I ��Һ�������mL�� |
4 |
8 |
15 |
20 |
50 |
120 |
150 |
|
|
C�������mL�� |
0 |
0 |
112 |
224 |
896 |
2240 |
2240 |
��12�֣�
��1��O=C=O��2�֣� ��2��Al(OH)3 HNO3 ����1�֣�
��3��4NH3+5O2
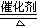 4NO+6H2O
��2�֣�
4NO+6H2O
��2�֣�
(4)4:3 ��2�֣� Al3++2 Na2O2= 4Na++ Al O2-+ O2����2�֣���5��1mol/L��2�֣�
��������