��Ŀ����
��14�֣��������ֿ���������A��B��C��D��E�� �������������������ӻ�����ͬ���ֱ������������� Na����Al3����Mg2����Ba2����Fe3�� ������������Cl����OH����NO3����CO32����X�е�һ�֡�
��1��ijͬѧͨ���ȽϷ�������Ϊ�������Ϳ��ж����б��е�����������
�� ���ѧʽ������Է�������С��д�ڵ�һ���գ���
��2��Ϊ��ȷ��X���ֽ���1���е��������ʼ�ΪA��B����X�����ʼ�C����C��B����Һ���ʱ���������ɫ��������ɫ��ζ���壻��C��A����Һ���ʱ�����ػ�ɫ������������ɫ������ó����е���ϡ������������ܽ⣬������а�ɫ���������ܽ⡣��XΪ ��
A��SO32�� B��SO42�� C��CH3COO�� D��SiO32��
��3��B��ˮ��Һ�������ԣ�ԭ��Ϊ �������ӷ���ʽ��ʾ����
��4����0. 02 molA��0.0 1molCͬʱ�ܽ�������������ˮ�У���ַ�Ӧ���������ó���������Ϊ
��������λ��Ч���֣���
��5����CuͶ�뵽װ��D��Һ���Թ��У�Cu���ܽ⣻�ٵμ�ϡH2SO4��Cu���ܽ⣬�ܿڸ����к���ɫ������֡�������Dһ���������������е� ������Ӧ�����ӷ��ţ����йط�Ӧ�����ӷ���ʽΪ�� ��
��6�����������Ѿ�ȷ�������ʣ����Լ����D��E�е������ӡ������ʵ��������衢������ ��
��1��Na2CO3��Ba(OH)2 ����1�֣���2�֣� ��2��B ��2�֣�
��3��CO32�� + H2O 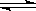 HCO3�� + OH����2�֣� ��4��6.1g ��2�֣�
HCO3�� + OH����2�֣� ��4��6.1g ��2�֣�
��5��NO3����2�֣��� 3Cu+8H++2NO3��=3Cu2++2NO��+4H2O��2�֣�
��6����D����Һ������Ba(OH)2��Һֱ�����������ȳ��ְ�ɫ�����������ܽ⣬��D�к���Al3+��E�к���Mg2+������D����Һ�м�������Na2CO3��Һ���������˰�ɫ��������ɫ��ζ�����壬��D�к���Al3+��E�к���Mg2+��������������Ҳ��2�֣�
��������
�����������1��̼�������ֻ���������ӽ�ϣ����һ������̼���ơ���ʱ����������ֻ���뱵���ӽ�ϣ�����һ������������������
��2����C��B����Һ���ʱ���������ɫ��������ɫ��ζ���壬���ɫ������������������ɫ��ζ������һ����CO2������B��̼���ƣ�C�к��������ӣ���C��A����Һ���ʱ�����ػ�ɫ������������ɫ������ó����е���ϡ������������ܽ⣬������а�ɫ���������ܽ⣬��˵��A������������C�л���������������C������������ѡB��
��3��̼������ǿ�������Σ�̼���ˮ����Һ�Լ��ԣ�����ʽΪCO32�� + H2O 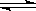 HCO3�� + OH����
HCO3�� + OH����
��4��0.02mol����������0.01mol�������������0.02mol���ᱵ��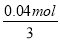 ������������������0.02mol��233g/mol+
������������������0.02mol��233g/mol+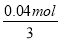 ��107g/mol��6.1g��
��107g/mol��6.1g��
��5����CuͶ�뵽װ��D��Һ���Թ��У�Cu���ܽ⣻�ٵμ�ϡH2SO4��Cu���ܽ⣬�ܿڸ����к���ɫ������֣�����ɫ������NO2����˵��D�к�������������������������������������ͭ����Ӧ�����ӷ���ʽΪ3Cu+8H++2NO3��=3Cu2++2NO��+4H2O��
��6��D�к�����������ӣ�E�к��������ӣ���ʣ���������ֻ�������Ӻ�þ���ӣ�������D��E�е������ӵ�ʵ������ǣ���D����Һ������Ba(OH)2��Һֱ�����������ȳ��ְ�ɫ�����������ܽ⣬��D�к���Al3+��E�к���Mg2+������D����Һ�м�������Na2CO3��Һ���������˰�ɫ��������ɫ��ζ�����壬��D�к���Al3+��E�к���Mg2+��
���㣺�������ӹ�������Ӽ�����йط��������

 ��������һ���þ�ϵ�д�
��������һ���þ�ϵ�д� Сѧ��10����Ӧ����ϵ�д�
Сѧ��10����Ӧ����ϵ�д��±���3�����ʵ��ܽ��(20 ��)������˵������ȷ����
���� | MgCl2 | Mg(OH)2 | MgCO3 |
�ܽ��(g/100 g) | 74 | 0.000 84 | 0.01 |
A����֪MgCO3��Ksp��6.82��10��6 mol2��L��2�������к��й���MgCO3����Һ�У�����c(Mg2��)��c(CO32-)����c(Mg2��)��c(CO32-)��6.82��10��6 mol2��L��2
B����ȥ�����к��е�MgCl2���ʣ���ѳ����Լ�ΪNa2CO3��Һ
C������������������ˮ��ϣ����ȡ����գ����յĹ��������ͬ
D����ʯ��ˮ��������Mg2����HCO3-��Ӳˮ�����������ӷ�Ӧ����ʽΪMg2����2HCO3-��Ca2����2OH����CaCO3����MgCO3����2H2O
 �� NH4Cl����ʽ��
�� NH4Cl����ʽ��
 �� ������ӵ����ģ�ͣ�
�� ������ӵ����ģ�ͣ�

