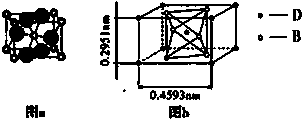
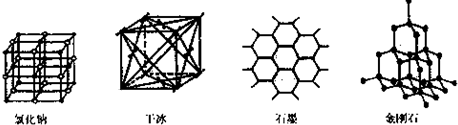
��Ŀ����
A��B��C��Ϊ��ѧ��ѧ�ij������ʣ��Һ�ͬһԪ�أ�XΪ���ʡ�����֮���ת����ϵ����ͼ��ʾ(��Ӧ���������������Ѿ���ȥ)![]()
(1)��A��B��C���³�ѹ�¾�Ϊ��̬�����C��ˮ��Ӧ����ǿ�ᣬ��A��B�Ļ�ѧ��Ӧ����ʽΪ_____________��Һ̬A�д��ڵ�������������(������)__________��
(2)��AΪ������Ԫ���γɵĵ��ʣ�C��ˮ��Ӧ����ǿ���C�ĵ���ʽ__________________�������Žϳ�ʱ���C�Ƿ���ʵļ�����____________��
(3)��XΪ��������C��ˮ��Һ�еμ�AgNO3��Һ���в�����ϡHNO3�İ�ɫ������������B�Ļ�ѧʽ��__________��C����Һ�ڱ���ʱӦ��������X��������____________________��
(4)��B�����ĺ�������������³���̬��c��ʹ��ɫʯ����Һ���ɫ������NaHCO3��Һ��Ӧ�ų�CO2����A��______________��C��NaHCO3��Ӧ�����ӷ���ʽΪ________________��
(1)4NH3+5O2![]() 4NO+6H2O
4NO+6H2O
���Ӽ�������(�»���) ��� ���ۼ�
(2) ![]() �۲�C����ɫ�Ƿ����ɵ���ɫ���ɫ�����У����ѱ���
�۲�C����ɫ�Ƿ����ɵ���ɫ���ɫ�����У����ѱ���
(3)FeCl3 Fe2+�ױ�������������Fe3+��Fe�ɽ�Fe3+��ԭΪFe2+
(4)�״�(��CH3OH)
HCOOH+![]() ====HCOO-+CO2��+H2O
====HCOO-+CO2��+H2O

��ϰ��ϵ�д�
�����Ŀ

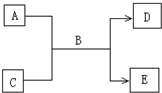 ��2009?�ϳ�ģ�⣩A��B��C��D��E��Ϊ��ѧ��ѧ�����Ĵ����BΪ��Ȼ���к�������Һ�壬����֮�������µķ�Ӧ��ϵ��
��2009?�ϳ�ģ�⣩A��B��C��D��E��Ϊ��ѧ��ѧ�����Ĵ����BΪ��Ȼ���к�������Һ�壬����֮�������µķ�Ӧ��ϵ��