��Ŀ����
��п��ZnS����һ����Ҫ�Ļ���ԭ�ϣ�������ˮ��������п�ķ���п����ȡt�乤����������ͼ��ʾ��
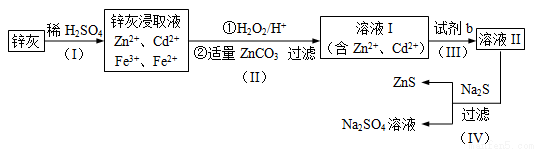
��1��Ϊ���п�ҵĽ�ȡ�ʣ��ɲ��õķ�����____________������ţ���
����ĥ �ڶ�ν�ȡ �������¶� �ܼ� ѹ �ݽ���
ѹ �ݽ���
��2����������������е�������_______________��д��ѧʽ����
��3��������пɵ�Cd���ʣ�Ϊ���������µ����ʣ��Լ�bӦΪ____________ ��
��4������������Ի���Na2SO4����ȡNa2S��
�ټ���ZnS�����Ƿ�ϴ�Ӹɾ��ķ�����______________��
��Na2S���ɵ����ʵ�����Na2S04��CH4�ڸ��¡�������������ȡ����ѧ��Ӧ��ʽΪ_________________��
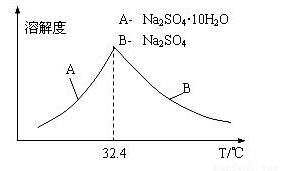
����֪Na2SO4��10H2O��Na2SO4���ܽ�����¶ȱ仯������ͼ������Һ�еõ�Na2SO4.10H2O�IJ���������________________________.
��5�������������ZnCO3Ϊb mol�����������CdΪd mol�����õ�VL�����ʵ���Ũ��Ϊc mol/L��Na2SO4��Һ��������������п���к���пԪ�ص�����Ϊ______________��
��ϰ��ϵ�д�
�����Ŀ


 ȷ����
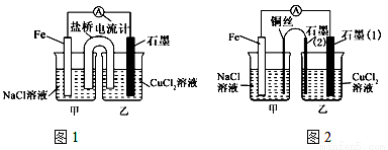
ȷ���� ���ҡ�S������ķ�Ӧһ�����Է�����
���ҡ�S������ķ�Ӧһ�����Է�����
 ��
�� B��
B��
 D��
D��