��Ŀ����
��ɫ����Һ�п��ܺ�����������:K+��Al3+��Fe3+��Ba2+��NO3����SO42����HCO32����Cl-,ȡ����Һ��������ʵ��:
������ɫʯ����ֽ������Һ,��ֽ�Ժ�ɫ;
��ȡԭ��Һ����,����ͭƬ��ϡ���Ṳ��,������ɫ����,������������������Ϊ����ɫ;
��ȡԭ��Һ����,���백ˮ�а�ɫ��������,�������������ˮ,��������ʧ;
��ȡԭ��Һ����,�����Ȼ�����Һ������ɫ����;
��ȡʵ��ܺ�ij�����Һ,������������Һ������ɫ����,�ټ��������ϡ����,��������ʧ��
��ش���������:
(1)��������ʵ���ж�ԭ��Һ���������ӿ϶����ڵ���������������������,�϶������ڵ����� ��
(2)д����ڢ�����ʵ���йص����ӷ���ʽ:
�� , ���� ��
������ɫʯ����ֽ������Һ,��ֽ�Ժ�ɫ;
��ȡԭ��Һ����,����ͭƬ��ϡ���Ṳ��,������ɫ����,������������������Ϊ����ɫ;
��ȡԭ��Һ����,���백ˮ�а�ɫ��������,�������������ˮ,��������ʧ;
��ȡԭ��Һ����,�����Ȼ�����Һ������ɫ����;
��ȡʵ��ܺ�ij�����Һ,������������Һ������ɫ����,�ټ��������ϡ����,��������ʧ��
��ش���������:
(1)��������ʵ���ж�ԭ��Һ���������ӿ϶����ڵ���������������������,�϶������ڵ����� ��
(2)д����ڢ�����ʵ���йص����ӷ���ʽ:
�� , ���� ��
(1)Al3+��NO3����SO42����Fe3+��Ba2+��HCO3��
(2)��3Cu+8H++2NO3�� 3Cu2++2NO��+4H2O
3Cu2++2NO��+4H2O
��Al3++3NH3��H2O Al(OH)3��+3NH4+
Al(OH)3��+3NH4+
(2)��3Cu+8H++2NO3��
 3Cu2++2NO��+4H2O
3Cu2++2NO��+4H2O��Al3++3NH3��H2O
 Al(OH)3��+3NH4+
Al(OH)3��+3NH4+������Һ��ɫ���ų���Fe3+�Ĵ���;�ɢٿ�֪��Һ������,�ų���HCO3���Ĵ���;�ɢڿ�֪NO3��һ������;�ɢۿ�֪Al3+����;�ɢܿ�֪S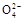 ����,��ô�ų���Ba2+�Ĵ���;���ڢ�������Cl-,���Ԣ���֤��ԭ��Һ���Ƿ�Cl-;���һ�����ڵ�������Al3+��NO3����SO42��,һ�������ڵ�������Fe3+��Ba2+��HCO3����
����,��ô�ų���Ba2+�Ĵ���;���ڢ�������Cl-,���Ԣ���֤��ԭ��Һ���Ƿ�Cl-;���һ�����ڵ�������Al3+��NO3����SO42��,һ�������ڵ�������Fe3+��Ba2+��HCO3����
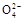 ����,��ô�ų���Ba2+�Ĵ���;���ڢ�������Cl-,���Ԣ���֤��ԭ��Һ���Ƿ�Cl-;���һ�����ڵ�������Al3+��NO3����SO42��,һ�������ڵ�������Fe3+��Ba2+��HCO3����
����,��ô�ų���Ba2+�Ĵ���;���ڢ�������Cl-,���Ԣ���֤��ԭ��Һ���Ƿ�Cl-;���һ�����ڵ�������Al3+��NO3����SO42��,һ�������ڵ�������Fe3+��Ba2+��HCO3����
��ϰ��ϵ�д�
 �Ƹ������������ϵ�д�
�Ƹ������������ϵ�д�
�����Ŀ
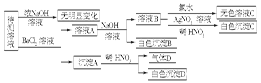
 2H++Cl��+ClO��
2H++Cl��+ClO��
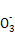 ��ClO-
��ClO-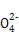 ��SCN-
��SCN-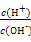 =1012����Һ��:N
=1012����Һ��:N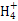 ��Al3+��S
��Al3+��S