��Ŀ����
���ֺ��ؽ��У�����ˮ��ȫռ�м�Ϊ��Ҫ�ĵ�λ��ij�о�С����ȡ��������Ⱦ��ˮԴ���з�����������������ʵ����Ϣ������һ������Ⱦ��ˮԴ����A��B�������ʣ�һ������C��D�������ʣ�һ������E���ʣ�A��B��C��D��E���ֳ��������ﶼ�����±��е������γɵģ�
Ϊ�˼�������������ֱ��������ʵ�飬�����ǣ�
�ٽ���������ˮ��DΪ��ɫ��Һ��������Ϊ��ɫ��Һ��
�ڽ�E��Һ���뵽C��Һ�У����ְ�ɫ�����������μӳ����ܽ⡣
�۽�����ɫ��Ӧʵ�飬ֻ��B��C���м����ӡ�
���ڸ���Һ�м���Ba(NO3)2��Һ���ټ������ϡ���ᣬA�зų���ɫ���壬C��D�в�����ɫ������
�ݽ�B��D����Һ��ϣ�δ���������������ɡ�
��������ʵ��������д���пհף�
(1)д��B��C��D�Ļ�ѧʽ��B________��C________��D________��
(2)����1 mol A����Һ�뺬1 mol E����Һ��Ӧ�����ɣ����õ�һ�ֻ�����û�����Ϊ________��
(3)д��ʵ��ڷ�����Ӧ�����ӷ���ʽ________________________________________
(4)C��������ˮ���������ӷ���ʽ��ʾ�侻ˮԭ��_______________________________
| ������ | K����Na����Cu2����Al3�� |
| ������ | SO42����HCO3����NO3����OH�� |
Ϊ�˼�������������ֱ��������ʵ�飬�����ǣ�
�ٽ���������ˮ��DΪ��ɫ��Һ��������Ϊ��ɫ��Һ��
�ڽ�E��Һ���뵽C��Һ�У����ְ�ɫ�����������μӳ����ܽ⡣
�۽�����ɫ��Ӧʵ�飬ֻ��B��C���м����ӡ�
���ڸ���Һ�м���Ba(NO3)2��Һ���ټ������ϡ���ᣬA�зų���ɫ���壬C��D�в�����ɫ������
�ݽ�B��D����Һ��ϣ�δ���������������ɡ�
��������ʵ��������д���пհף�
(1)д��B��C��D�Ļ�ѧʽ��B________��C________��D________��
(2)����1 mol A����Һ�뺬1 mol E����Һ��Ӧ�����ɣ����õ�һ�ֻ�����û�����Ϊ________��
(3)д��ʵ��ڷ�����Ӧ�����ӷ���ʽ________________________________________
(4)C��������ˮ���������ӷ���ʽ��ʾ�侻ˮԭ��_______________________________
(1)KNO3��KAl(SO4)2��CuSO4��(2)Na2CO3
(3)Al3����3OH��=Al(OH)3����Al(OH)3��OH��=AlO2����2H2O
(4)Al3����3H2O=Al(OH)3(����)��3H��
(3)Al3����3OH��=Al(OH)3����Al(OH)3��OH��=AlO2����2H2O
(4)Al3����3H2O=Al(OH)3(����)��3H��
������ɿ�֪��A��B��C��D���ܴ������档�ɢ�֪��D��Һ�к�Cu2������C�в���OH����HCO3�����ɢ�֪��E��Һ�к�OH����C��Һ�к�Al3�����ɢ�֪��B��C��Һ�к��м����ӣ�A��D��E�в��������ӡ��ɢ�֪��A�к�HCO3����C��D��Һ�к�SO42����B��E�в���SO42�����ɢݿ�֪��B�в���OH����HCO3��������������
DΪCuSO4 , CΪKAl(SO4)2��EΪNaOH ��BΪKNO3��AΪNaHCO3��
DΪCuSO4 , CΪKAl(SO4)2��EΪNaOH ��BΪKNO3��AΪNaHCO3��

��ϰ��ϵ�д�
�����Ŀ

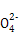
 ��Ba2+��OH-��I-
��Ba2+��OH-��I-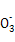
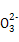 ��Cl-
��Cl-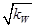 mol/L����Һ�У�K����Fe3����Cl����CO32��
mol/L����Һ�У�K����Fe3����Cl����CO32��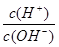 ��1��10��12����Һ��K����AlO2����CO32����Na��
��1��10��12����Һ��K����AlO2����CO32����Na�� ��S
��S ��Si
��Si
 H++OH-��KW=10-14
H++OH-��KW=10-14 b(����ڡ���С�ڡ����ڡ�)����a��b��ʾNH3��H2O�ĵ���ƽ�ⳣ��Ϊ����������
b(����ڡ���С�ڡ����ڡ�)����a��b��ʾNH3��H2O�ĵ���ƽ�ⳣ��Ϊ����������