��Ŀ����
2������٤������ԼΪ6.02��1023mol-1��������������ȷ���ǣ�������| A�� | ��״���£�4.48LCH3CH2OH��Լ����1.204��1023���ǻ� | |
| B�� | ���³�ѹ�£�31g����P4�ͺ���P8�Ļ����Լ����6.02��1023����ԭ�� | |
| C�� | ��״���£�7.8g����Լ����1.806��1023��̼̼˫�� | |
| D�� | 1molH2O2��MnO2����������ȫ��Ӧת�Ƶĵ�����ԼΪ1.204��1024������ |
���� A������Ħ��������ö���Ϊ���壻
B�����ͺ����Ӷ�������ԭ�ӹ��ɣ�����N=$\frac{m}{M}$��NA���㣻
C�������Ӳ�����̼̼˫����
D��˫��ˮ�ֽ��У�˫��ˮ�������������ǻ�ԭ��������1mol����ת��2mol���ӣ�
��� �⣺A������£��Ҵ���Һ̬������ʹ������Ħ���������A����
B�����ͺ����Ӷ�������ԭ�ӹ��ɣ�31g����P4�ͺ���P8�Ļ���ﺬ����ԭ�Ӹ���Ϊ��$\frac{31g}{31g/mol}$��NA=NA��Լ����6.02��1023����ԭ�ӣ���B��ȷ��
C�������Ӳ�����̼̼˫������C����
D.1molH2O2��MnO2����������ȫ��Ӧ����0.5mol������ע��1mol���ӣ�ת�Ƶĵ�����ԼΪ6.02��1023�����ӣ���D����
��ѡ��B��
���� ���⿼���˰���٤���������йؼ��㣬����������ع�ʽ�ǽ���ؼ���ע�ⱽ�Ľṹ��ע������Ħ����������ö�����Ŀ�ѶȲ���

��ϰ��ϵ�д�
�����Ŀ
5���ӷ�������ԾȵȰ�ȫ֪ʶ�жϣ�����������ȷ���ǣ�������
| A�� | �����Ż����Ͽ��Ŵ� | B�� | ����ʱ���Ż��ù��Ǹ��� | ||
| C�� | ���������ȼ���Ƿ�й© | D�� | �����Ż�ʱ��ˮ���� |
13��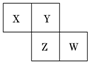 X��Y��Z��W��Ϊ������Ԫ�أ����������ڱ������λ����ͼ��ʾ����Yԭ�ӵ��������������ڲ��������3��������˵������ȷ���ǣ�������
X��Y��Z��W��Ϊ������Ԫ�أ����������ڱ������λ����ͼ��ʾ����Yԭ�ӵ��������������ڲ��������3��������˵������ȷ���ǣ�������
 X��Y��Z��W��Ϊ������Ԫ�أ����������ڱ������λ����ͼ��ʾ����Yԭ�ӵ��������������ڲ��������3��������˵������ȷ���ǣ�������
X��Y��Z��W��Ϊ������Ԫ�أ����������ڱ������λ����ͼ��ʾ����Yԭ�ӵ��������������ڲ��������3��������˵������ȷ���ǣ�������| A�� | ����������Ӧˮ���������W��Z�� | |
| B�� | ֻ��������Ԫ�ز�������л������� | |
| C�� | Z�ĵ�����������Ӧ��Y���� | |
| D�� | X��Y�γɵĻ����ﶼ������ˮ |
10�� Ԫ��R��X��T��Z��Q��Ԫ�����ڱ��е����λ�����±���ʾ������R�����ڰ�����H2���һ��ϲ�������ը���������ж���ȷ���ǣ�������
Ԫ��R��X��T��Z��Q��Ԫ�����ڱ��е����λ�����±���ʾ������R�����ڰ�����H2���һ��ϲ�������ը���������ж���ȷ���ǣ�������
 Ԫ��R��X��T��Z��Q��Ԫ�����ڱ��е����λ�����±���ʾ������R�����ڰ�����H2���һ��ϲ�������ը���������ж���ȷ���ǣ�������
Ԫ��R��X��T��Z��Q��Ԫ�����ڱ��е����λ�����±���ʾ������R�����ڰ�����H2���һ��ϲ�������ը���������ж���ȷ���ǣ�������| A�� | �ǽ����ԣ�Z��T��X | B�� | R��Q�ĵ��������26 | ||
| C�� | ��̬�⻯���ȶ��ԣ�R��T��Q | D�� | ����������ˮ��������ԣ�T��Q |
17������˵����ȷ���ǣ�������
| A�� | ��ά�غ͵�������ˮ������ɫ | |
| B�� | ��֬������������� | |
| C�� | ʯ�ͷ���õ��������Ǵ����� | |
| D�� | ú���������������仯��ú�ĸ������ڻ�ѧ�仯 |
14����ѧѧ����Ҫ������ѧר�����������������йػ�ѧ������ȷ���ǣ�������
| A�� | CO2���ӱ���ģ�ͣ� | B�� | NH4I�ĵ���ʽ��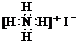 | ||
| C�� | �Ҵ��Ľṹ��ʽ��C2H5OH | D�� | K+���������Ų�ʽ��4S24P6 |
11��һ���¶��£���Ũ��Ϊ0.1mol•L-1��Na2CO3��Һ��ˮ����ϡ�ͣ����и�ֵʼ��������ǣ�������
| A�� | c��H+�� | B�� | c��HCO${\;}_{3}^{-}$�� | ||
| C�� | c��H+��•c��OH-�� | D�� | $\frac{c��O{H}^{-}��•c��HC{{O}_{3}}^{-}��}{c��C{{O}_{3}}^{2-}��}$ |
12�����·�Ӧʽ����ȷ���ǣ�������
| A�� | ������Ͷ��ˮ�е����ӷ���ʽ��Na+2H2O=Na++2OH-+O2�� | |
| B�� | �����£�0.1mol/L�Ȼ����ҺpH=5��ԭ��NH4++H2O?NH3•H2O+H+ | |
| C�� | �ö��Ե缫��ⱥ��ʳ��ˮ��������Ӧʽ��2Cl--2e-=Cl2�� | |
| D�� | ��ȩ�����Ƶ�������ͭ����Һ��Ӧ�����ӷ���ʽ��CH3CHO+2Cu��OH��2+OH-$\stackrel{��}{��}$CH3COO-+Cu2O��+3H2O |
 ���ܶ�����л�����E���¿���H2�����ӳɷ�Ӧ�������ϩ����H��Ȳ���۱����ܼ�ȩ������̼ԭ�Ӳ�ȡsp2�ӻ��ķ����Т٢ۢܣ���������ţ���HCHO���ӵ�����ṹΪƽ�������Σ����ӳɲ�����ۡ��е��CH4���ۡ��е�ߣ�����Ҫԭ���Ǽӳɲ���CH3OH����֮�����γ��������ָ���ӳɲ����Ǻ����ʣ�
���ܶ�����л�����E���¿���H2�����ӳɷ�Ӧ�������ϩ����H��Ȳ���۱����ܼ�ȩ������̼ԭ�Ӳ�ȡsp2�ӻ��ķ����Т٢ۢܣ���������ţ���HCHO���ӵ�����ṹΪƽ�������Σ����ӳɲ�����ۡ��е��CH4���ۡ��е�ߣ�����Ҫԭ���Ǽӳɲ���CH3OH����֮�����γ��������ָ���ӳɲ����Ǻ����ʣ�