��Ŀ����
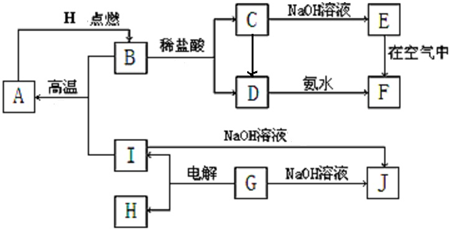
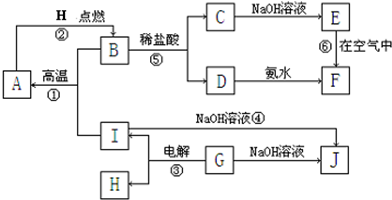
A��I�ֱ��ʾ��ѧ��ѧ�г�����һ�����ʣ�����֮�����ϵ����ͼ��ʾ�����ַ�Ӧ�������û���г���������֪GΪ����Ԫ�صĹ�̬�����A��B��C��D��E��F���������о���ͬһ��Ԫ�ء�
����д���пհף�
��1��A��B��C��D��E��F��������������ͬһ��Ԫ�������ڱ���λ�õ�__ _���ڡ���_ __�� ��
��2��д����Ӧ�ٵĻ�ѧ����ʽ___________ _________��
��3��д��D��J��ˮ��Һ��Ӧ�����ӷ���ʽ_______ ___��
��4��д����Ӧ�۵ĵ缫����ʽ�������� ��
������ ��
��5���������仯�ĽǶȿ����٢ڢ۷�Ӧ�����ڡ�H��0�ķ�Ӧ��___________��������ţ�
��6����Ӧ���е���������_____________________��
��7��B�Ļ�ѧʽ��_______________��H�Ľṹʽ����������������������
��14�֣���1���������ڡ��ڢ��壨ÿ��1�֣���2�֣�
��2��8Al��3Fe3O4![]() 4Al2O3��9Fe��2�֣��������Բ����֣�
4Al2O3��9Fe��2�֣��������Բ����֣�
��3��Fe3++3AlO2-+6H2O=Fe(OH)3+3Al(OH)3��2�֣�
��4��������6O2����12e����3O2����������4Al3+��12e����4Al
��ÿ��2�֣���4�֡�����������Ҳ�ɸ��֣�
��5���٢ڣ�1�֣���ѡ��ѡ�����֣�
��6��ˮ(д����ʽҲ�ɣ���1�֣�
��7��Fe3O4 O=O��ÿ��1�֣���2�֣�

 �Ķ��쳵ϵ�д�
�Ķ��쳵ϵ�д�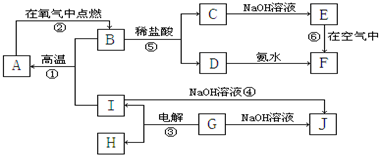


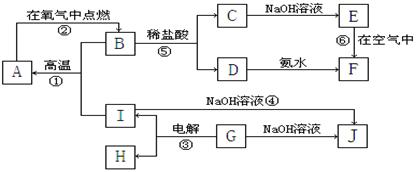
 A��I�ֱ��ʾ��ѧ��ѧ�г�����һ�����ʣ�����֮���ת����ϵ��ͼ��ʾ�����ַ�Ӧ�������û���г���������֪A��B��C��D��E��F���������о���ͬһ��Ԫ�أ�A��I�����ֳ����Ľ������ʣ�H�ڳ�������һ�ֳ����ķǽ�����̬���ʣ�
A��I�ֱ��ʾ��ѧ��ѧ�г�����һ�����ʣ�����֮���ת����ϵ��ͼ��ʾ�����ַ�Ӧ�������û���г���������֪A��B��C��D��E��F���������о���ͬһ��Ԫ�أ�A��I�����ֳ����Ľ������ʣ�H�ڳ�������һ�ֳ����ķǽ�����̬���ʣ�