��Ŀ����
���Ų��Ͽ�ѧ�ķ�չ�����������仯����õ���Խ��Խ�㷺��Ӧ�ã�������Ϊ���Ͻ�ά���ء�����ҵ�ϻ��շϷ�����������V2O5��VOSO4��K2SO4��SiO2���з�����Ҫ�������£�
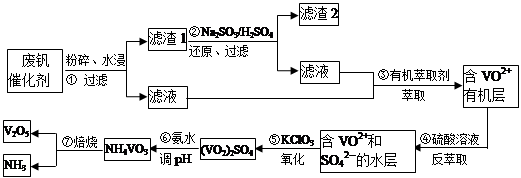
��֪����1��V2O5��NH4VO3��Ϊ�����VOSO4��(VO2)2SO4��Ϊ�����
��2�� 2VO2++H2C2O4+2H+ = 2VO2+ + 2CO2��+ 2H2O
�ش��������⣺
��1�������ǰ�������Ŀ����_________________________��
��2��������з�����Ӧ�����ӷ���ʽΪ__________________________��
��3������۵ı仯���̿ɼ�Ϊ(HA��ʾ�л���ȡ��)��
VOSO4 (ˮ��)+ 2HA���л��㣩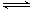 VOA2(�л��㣩+ H2SO4(ˮ��)��������п�ѡ������������ȡ��ԭ����_____________��
VOA2(�л��㣩+ H2SO4(ˮ��)��������п�ѡ������������ȡ��ԭ����_____________��
��4���������ữ��H2C2O4��Һ�ζ�(VO2)2SO4��Һ���Բⶨ�����ݺ���Һ�к������IJ���Ϊ��ȡ10.0mL0.1mol/LH2C2O4��Һ����ƿ�У�����ָ�����������Һʢ���ڵζ����У��ζ����յ�ʱ�����Ĵ���Һ�����Ϊ10.0mL���ɴ˿�֪(VO2)2SO4��Һ��Ԫ�صĺ���Ϊ_________g/L��
��5��V2O5���ý���(��Ca��Al)�Ȼ�ԭ����÷�����������Ȼ�ԭ�Ƶ÷��Ļ�ѧ����ʽΪ_______________��

��֪����1��V2O5��NH4VO3��Ϊ�����VOSO4��(VO2)2SO4��Ϊ�����
��2�� 2VO2++H2C2O4+2H+ = 2VO2+ + 2CO2��+ 2H2O
�ش��������⣺
��1�������ǰ�������Ŀ����_________________________��
��2��������з�����Ӧ�����ӷ���ʽΪ__________________________��
��3������۵ı仯���̿ɼ�Ϊ(HA��ʾ�л���ȡ��)��
VOSO4 (ˮ��)+ 2HA���л��㣩
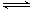 VOA2(�л��㣩+ H2SO4(ˮ��)��������п�ѡ������������ȡ��ԭ����_____________��
VOA2(�л��㣩+ H2SO4(ˮ��)��������п�ѡ������������ȡ��ԭ����_____________����4���������ữ��H2C2O4��Һ�ζ�(VO2)2SO4��Һ���Բⶨ�����ݺ���Һ�к������IJ���Ϊ��ȡ10.0mL0.1mol/LH2C2O4��Һ����ƿ�У�����ָ�����������Һʢ���ڵζ����У��ζ����յ�ʱ�����Ĵ���Һ�����Ϊ10.0mL���ɴ˿�֪(VO2)2SO4��Һ��Ԫ�صĺ���Ϊ_________g/L��
��5��V2O5���ý���(��Ca��Al)�Ȼ�ԭ����÷�����������Ȼ�ԭ�Ƶ÷��Ļ�ѧ����ʽΪ_______________��
��1�������Һ�Ӵ�������ӿ�������ʣ���߽����ʣ�2�֣�
��2��V2O5+ SO32��+4H+=2VO2+ + SO42��+2H2O��2�֣�
��3���������ᣬ��ʹƽ��������У�ʹVOSO4����ˮ�㣻��2�֣�
��4��10.2g/L ��2�֣�
��5��3V2O5+10Al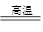 6V+5Al2O3��2�֣�
6V+5Al2O3��2�֣�
��2��V2O5+ SO32��+4H+=2VO2+ + SO42��+2H2O��2�֣�
��3���������ᣬ��ʹƽ��������У�ʹVOSO4����ˮ�㣻��2�֣�
��4��10.2g/L ��2�֣�
��5��3V2O5+10Al
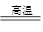 6V+5Al2O3��2�֣�
6V+5Al2O3��2�֣������������1��������巴Ӧ����������Һ�Ӵ�������ӿ�������ʣ���߽����ʡ�
��2������������л�ԭ�ԣ����������£��ܱ���������������������������ӣ����ӷ�Ӧ����ʽΪ��V2O5+ SO32��+4H+=2VO2+ + SO42��+2H2O
��3������H2SO4��H2SO4Ũ��������ۻ�ѧƽ���������ƶ���ʹVOSO4����ˮ�㡣
��4��������Ŀ������Ϣ��2VO2+ ~ H2C2O4����֪(VO2)2SO4��Һ��Ԫ�صĺ���Ϊ��0.1mol/L��0.01L��2��51g/mol��0.01L= 10.2g/L��
��5������������������Ӧ���ɷ�������������ѧ����ʽΪ��3V2O5+10Al
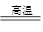 6V+5Al2O3
6V+5Al2O3
��ϰ��ϵ�д�
 ��У����ϵ�д�
��У����ϵ�д�
�����Ŀ
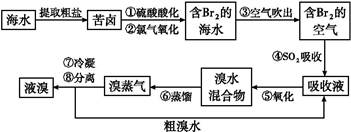
 2H++2Br-+S
2H++2Br-+S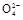
 Ti+2MgCl2��ú���״�ѡ��÷�Ӧ���������ֻ����н���_______ (�����)��
Ti+2MgCl2��ú���״�ѡ��÷�Ӧ���������ֻ����н���_______ (�����)��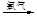
 ������
������
 Һ��
Һ�� 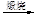
 ʯ����
ʯ����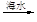
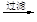
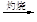 MgO
MgO þ
þ
 �����л���Һ
�����л���Һ
 ����
����