��Ŀ����
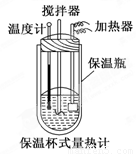
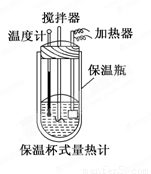
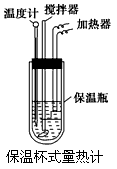
�����ȼ���(��ͼ)��100 mL 0.50 mol��L��1��CH3COOH��Һ��100 mL 0.55 mol��L��1��NaOH��Һ��ϣ��¶ȴ�298.0 K������300.7 K����֪���ȼƵ����ݳ���(���ȼƸ�����ÿ����1 K����Ҫ����)��150.5 J��K��1����Һ�ܶȾ�Ϊ1 g��mL��1��������Һ�ı�����c��4.184 J��(g��K)��1��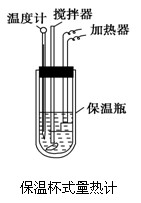
(1).����CH3COOH���к��Ȧ�H������ֵʽ��
(2).������ֵ�����57.3 kJ/mol��ƫ�����ԭ�������
a��ʵ��װ�ñ��¡�����Ч����
b������0.55 mol/L NaOH��Һʱ���ӿ̶��߶���
c���ֶ�ΰ�NaOH��Һ����ʢ�������С�ձ���
d�����¶ȼƲⶨNaOH��Һ��ʼ�¶Ⱥ�ֱ�ӲⶨH2SO4��Һ���¶�
e������Ͳ��ȡNaOH��Һ�����ʱ���Ӷ���
(1) CH3COOH���к��ȣ���H����53.3 kJ��mol��1��
��2��a c d����ѡ����ѡ�������֣�
����

��ϰ��ϵ�д�
 ��У����ϵ�д�
��У����ϵ�д�
�����Ŀ