��Ŀ����
�±�ΪԪ�����ڱ���һ���֣������Ԫ�آ٣����ڱ��е�λ�ã��û�ѧ����ش���������
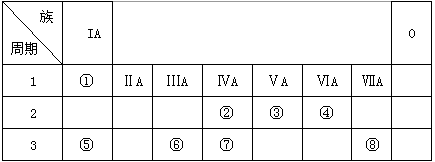
��1���ܡ��ݡ���ԭ�Ӱ뾶�ɴ�С��˳��Ϊ_________________________��
��2���ڡ��ۡ��ߵ���ۺ������������ǿ������˳����_________________________��
��3���١��ܡ��ݡ����е�ijЩԪ�ؿ��γɼȺ����Ӽ��ֺ����Թ��ۼ��Ļ����д������һ�ֻ�����ĵ���ʽ��____________________
��4���ɱ�������Ԫ�ص�ԭ�Ӱ�1��1��ɵij���Һ̬�������ϡҺ�ױ����ֽ⣬��ʹ�õĴ���Ϊ������ţ�_________________��
a��MnO2 b��FeCl3 c��Na2SO3 d��KMnO4
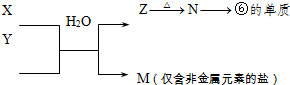
��5���ɱ���Ԫ���γɵij�������X��Y��Z��M��N�ɷ������·�Ӧ��
��2���ڡ��ۡ��ߵ���ۺ������������ǿ������˳����_________________________��
��3���١��ܡ��ݡ����е�ijЩԪ�ؿ��γɼȺ����Ӽ��ֺ����Թ��ۼ��Ļ����д������һ�ֻ�����ĵ���ʽ��____________________
��4���ɱ�������Ԫ�ص�ԭ�Ӱ�1��1��ɵij���Һ̬�������ϡҺ�ױ����ֽ⣬��ʹ�õĴ���Ϊ������ţ�_________________��
a��MnO2 b��FeCl3 c��Na2SO3 d��KMnO4
��5���ɱ���Ԫ���γɵij�������X��Y��Z��M��N�ɷ������·�Ӧ��

X��Һ��Y��Һ��Ӧ�����ӷ���ʽΪ_____________________��N���ĵ��ʵĻ�ѧ����ʽΪ________________�������£�Ϊʹ0��1 mol/L M ��Һ����M���������������Ũ����ȣ�Ӧ����Һ�м���һ������Y��Һ��_________________��
��1��Na>Al>O
��2��HNO3>H2CO3>H2SiO3
��3��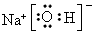 ��
��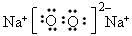
��4��a��b
��5��Al3++3NH3��H2O==Al(OH)3��+3NH4+��2Al2O3(����) 4Al+3O2������ҺpH����7
4Al+3O2������ҺpH����7

��ϰ��ϵ�д�
 ��������һ���þ�ϵ�д�
��������һ���þ�ϵ�д� Сѧ��10����Ӧ����ϵ�д�
Сѧ��10����Ӧ����ϵ�д�
�����Ŀ








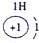
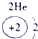
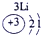
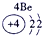
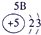
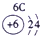
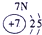
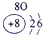
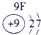
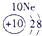
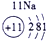





 ��ʾ����
��ʾ����