��Ŀ����
A��B��C��D���ֶ�����Ԫ�ص�ԭ��������������A��Dͬ�壻B��Cͬ���ڡ�A��B��ɵĻ������Ϊ��̬������A��Bԭ�Ӹ���֮��Ϊ4��1�������£���A��C��ɵ����ֻ������Һͱ���ΪҺ̬������A��Cԭ�Ӹ�����Ϊ1��1������A��Cԭ�Ӹ�����Ϊ2��1����D��C��ɵ����ֻ����ﶡ���춼Ϊ��̬������D��Cԭ�Ӹ���֮��Ϊ1��1������D��Cԭ�Ӹ�����2��1��BԪ�ص��������������ܷ�����ѧ��Ӧ��������Ϊ���֣�����һ��ΪC�ĵ��ʡ�(1)�����ʵĻ�ѧʽ�ֱ�Ϊ����________________����___________����________________����________________����________________��
(2)BԪ�ص���������������Ӧ�Ļ�ѧ����ʽΪ_________________________________��
��������ԭ�Ӹ�����Ϊ4��1�Ļ�����ΪCH4����AΪH��BΪC����HԪ���γɵ�ԭ�Ӹ�����Ϊ1��1��2��1��Һ̬������ΪH2O2��H2O����CΪO����ΪH2O2����ΪH2O������Ԫ���γɵĹ�̬������ΪNa2O2��Na2O����DΪNa����ΪNa2O2����ΪNa2O��
�𰸣�(1)CH4 H2O2 H2O Na2O2 Na2O
(2)2CO2+2Na2O2![]() 2Na2CO3+O2
2Na2CO3+O2

��ϰ��ϵ�д�
�����Ŀ
A��B��C��D���ֶ�����Ԫ�ص�ԭ�Ӱ뾶���μ�С��A��C�ĺ˵����֮��3��4��D�ֱ���A��B��C���γ�10�����ӵķ���X��Y��Z��������������ȷ���ǣ�������
| A��X��Y��Z���ַ��ӵ��ȶ������� | B����������Ԫ�ع����γ����ֵ��� | C��X��Y��Z���ַ��ӵķе������� | D��X��Y��Z���ַ��Ӿ�Ϊ���Է��� |

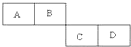 A��B��C��D���ֶ�����Ԫ�������ڱ��е����λ�������ʾ��A�ĵ�����ˮ������Ӧ������ȡˮú����
A��B��C��D���ֶ�����Ԫ�������ڱ��е����λ�������ʾ��A�ĵ�����ˮ������Ӧ������ȡˮú����