��Ŀ����
ʵ������Ҫ1.0 mol��L-1NaOH��Һ400 mL��������Һ����������ش��������⣺
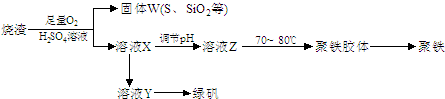
��1�� ��ͼ��ʾ��������������Һ�϶�����Ҫ���� ������ţ�������������Һ�����õ��IJ��������� �����������ƣ���
 |
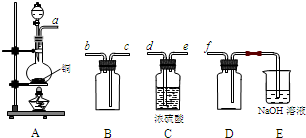
��2�������й�����ƿ�Ĺ����ʹ�������У���ȷ���� ������ţ���
A����������һ������ġ�Ũ��ȷ����Һ
B������������Һ
C����������ƿ������µ����������Һ��
D������ƿ�ϱ����¶Ⱥ��ݻ�
E������һ�������Ũ�ȵ���Һʱ������ƿδ���������ҺŨ��ƫС
��3�����������������ƽ��ȡ����NaOH������Ϊ g����400mL���Ƶ���Һ��ͨ��0.3molCO2����������Һ�����ʵĻ�ѧʽΪ ��
��1��A B ��2�֣� ������ ��1�֣� ��2��A D ��2�֣�
��3��20.0 ��2�֣� Na2CO3 NaHCO3��2�֣�

��ϰ��ϵ�д�
�����Ŀ
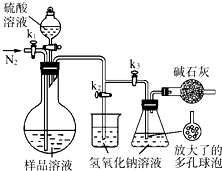 ��2013?������ģ��̼����-��������Ӻ��aNa2CO3?bH2O2������Ư�ס�ɱ�����ã�ʵ�����á����������Ʊ������ʵ�ʵ�鲽�����£�
��2013?������ģ��̼����-��������Ӻ��aNa2CO3?bH2O2������Ư�ס�ɱ�����ã�ʵ�����á����������Ʊ������ʵ�ʵ�鲽�����£�
