��Ŀ����
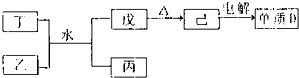
����������Ԫ��A��B��C��D��ԭ������������������A��Cͬ���壬B��C��Dͬ���ڣ�Aԭ�ӵ������������Ǵ�����������3����B�Ƕ�����Ԫ����ԭ�Ӱ뾶��������Ԫ�ء�������������⣺?��1��A��Ԫ�ط��ţ���D��ԭ�ӽṹʾ��ͼ��������������������������?
��2��A��B��C����Ԫ���γɵļ����ӵİ뾶�ɴ�С��˳������������������
��3��A��B��C��D�γɵĻ�����B2A2��CD2��D2A��DA2�У���ԭ�Ӷ����������8���ӽṹ����
����������������������д����Ļ�ѧʽ����?
��4��CA2��DԪ�صĵ�����ˮ��Һ�з�Ӧ�Ļ�ѧ����ʽ����������������������?
�����������ͻ�ƿ�����Aԭ�ӵ����������Ǵ�����3����˵�������һ����L�㣬�����һ����K�㡣��˸�Ԫ��Ϊ��Ԫ�ء�CԪ��Ӧ��ΪS,�Դ�Ϊͻ�ƣ����Եõ���Ӧ�𰸡�
�𰸣�![]() ???
???
��2��S2����O2����Na+??
��3��Na2O2��SCl2��Cl2O?
��4��SO2+Cl2+2H2O![]() 2HCl+H2SO4
2HCl+H2SO4

��ϰ��ϵ�д�
 �Ķ��쳵ϵ�д�
�Ķ��쳵ϵ�д�
�����Ŀ
����������Ԫ��A��B��C��D��ԭ��������������A��Cԭ���������8��A��B��C����Ԫ��ԭ�ӵ�����������֮��Ϊ15��Bԭ����������������Aԭ��������������һ�룮����������ȷ���ǣ�������
| A��ԭ�Ӱ뾶��A��D��C��B | B��B��C��D�ֱ���A�γɵĻ�����һ��������ͬ�Ļ�ѧ�� | C������������Ӧˮ��������ԣ�D��C | D�������£�����B�ܴ�������Ũ������ |
����������Ԫ��A��B��C��ԭ���������ε�����A��Cͬ���壬Bԭ�ӵ���������������Aԭ�ӵĴ���������������ԭ�ӵ�����������֮��Ϊ10��������������ȷ���ǣ�������
| A��ԭ�ڰ뾶A��B��C | B��A����̬�⻯���ȶ��Դ���C����̬�⻯���ȶ��� | C��A��C��Ԫ����������������ˮ���ϵõ���Ӧ���� | D������ʱ��A���ʿ��Դ�C�����������û��õ�C���� |
����������Ԫ��A��B��C��D��E��ԭ������������������A��Cͬ���壬A������Ԫ�ز���ͬһ���ڣ�B��Dͬ���壬������D�ĵ���Ϊ����ɫ���壮�����ƶ�����ȷ���ǣ�������
| A��ԭ�Ӱ뾶��С�����˳��r��C����r��D����r��E�� | B��Ԫ��D��E�ֱ���A�γɵĻ���������ȶ��ԣ�E��D | C��Ԫ��D������������Ӧˮ��������Ա�E��ǿ | D��Ԫ��B�ֱ���A��C�γɵĻ������л�ѧ����������ȫ��ͬ |
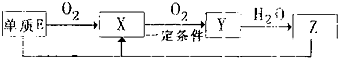
 ����������Ԫ��A��B��C��D��E��ԭ������������������ԭ�Ӻ���ĵ��Ӳ���֮��Ϊ10��BԪ�صĻ���������࣬��Ŀ�Ӵ�C��D����Ԫ���γɵĵ����ǿ����к����������ʣ�D��E��Ԫ�ؿ����������ֲ�ͬ�����ӻ����
����������Ԫ��A��B��C��D��E��ԭ������������������ԭ�Ӻ���ĵ��Ӳ���֮��Ϊ10��BԪ�صĻ���������࣬��Ŀ�Ӵ�C��D����Ԫ���γɵĵ����ǿ����к����������ʣ�D��E��Ԫ�ؿ����������ֲ�ͬ�����ӻ���� NH3?H2O+H+
NH3?H2O+H+ 2NO2
2NO2