��Ŀ����
Ԫ�����ڱ���ѧϰ��ѧ����Ҫ���ߣ���������������Ϣ���ɣ������ǰ��ֶ�����Ԫ�ص������Ϣ����֪���ԭ�Ӱ뾶Ϊ0.089nm��
Fԭ���������ӣ�G����������븺������ȣ��������������Ǵ����Ķ�����HԪ�ص�����ɫ��Ӧ�ʻ�ɫ
��1��BԪ����Ԫ�����ڱ��е�λ��
��2����������Ԫ�ص�����������Ӧ��ˮ������������ǿ����
��3���õ���ʽ��ʾA��D�γɻ�����Ĺ��̣�
 ��
��
��4��H��E�γ�ԭ�Ӹ�����Ϊ1��1�Ļ�����Ϊ
��5��F��G�γɵ���Ļ�����ĵ���ʽ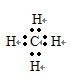
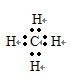 ���÷��ӵĿռ乹��Ϊ
���÷��ӵĿռ乹��Ϊ
��6��E���⻯���C���⻯����۷е�ߵ�ԭ����
��7�����б���ʳ��ˮ��ˮ���У���һ������ˮ���е��Թ����ű���ʳ��ˮ���Ⱥ��ռ��������GF4��D�ĵ������壬�ڹ��������¿ɹ۲쵽ˮ�����о����������Թ���������ɫ��dz��Һ��������
| Ԫ�ش��� | A | B | C | D | E |
| ԭ�Ӱ뾶/nm | 0.160 | 0.143 | 0.102 | 0.099 | 0.074 |
| ��Ҫ���ϼ� | +2 | +3 | +6��-2 | -1 | -2 |
��1��BԪ����Ԫ�����ڱ��е�λ��
�������ڵڢ�A��
�������ڵڢ�A��
����2����������Ԫ�ص�����������Ӧ��ˮ������������ǿ����
HClO4
HClO4
���ѧʽ������3���õ���ʽ��ʾA��D�γɻ�����Ĺ��̣�


��4��H��E�γ�ԭ�Ӹ�����Ϊ1��1�Ļ�����Ϊ
Na2O2
Na2O2
���ѧʽ�������еĻ�ѧ������Ϊ���Ӽ��ͷǼ��Լ�
���Ӽ��ͷǼ��Լ�
����5��F��G�γɵ���Ļ�����ĵ���ʽ
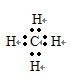
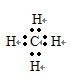
��������
��������
����6��E���⻯���C���⻯����۷е�ߵ�ԭ����
ˮ���Ӽ�������
ˮ���Ӽ�������
����7�����б���ʳ��ˮ��ˮ���У���һ������ˮ���е��Թ����ű���ʳ��ˮ���Ⱥ��ռ��������GF4��D�ĵ������壬�ڹ��������¿ɹ۲쵽ˮ�����о����������Թ���������ɫ��dz��Һ��������
�Թܱڳ�����״Һ��
�Թܱڳ�����״Һ��
���Թ�������������
�Թ�������������
�����������������Fԭ���������ӣ�������H��G����������븺������ȣ����������������Ǵ����Ķ���������G��C��HԪ�ص�����ɫ��Ӧ�ʻ�ɫ��ӦΪNa��A��Mg��B��Al��C��S��D��Cl��E��O
��1������Ԫ�������ڱ��е�λ�����ش�
��2��ͬ���ڴ���������������Ӧˮ�������������ǿ��ͬ������ϵ�������������Ӧˮ�����������������
��3���Ȼ�þ��þ���Ӻ�������֮��ͨ�����Ӽ��γɵ����ӻ����
��4�����ݹ������ƵĻ�ѧ���������ش�
��5����������������ṹ�ķ��ӣ��ǹ��ۻ����
��6��ˮ���Ӽ�������������ˮ�ķе�ϸߣ�
��7������������ڹ��յ������·���ȡ����Ӧ���������������л����һ�����
��1������Ԫ�������ڱ��е�λ�����ش�
��2��ͬ���ڴ���������������Ӧˮ�������������ǿ��ͬ������ϵ�������������Ӧˮ�����������������
��3���Ȼ�þ��þ���Ӻ�������֮��ͨ�����Ӽ��γɵ����ӻ����
��4�����ݹ������ƵĻ�ѧ���������ش�
��5����������������ṹ�ķ��ӣ��ǹ��ۻ����
��6��ˮ���Ӽ�������������ˮ�ķе�ϸߣ�
��7������������ڹ��յ������·���ȡ����Ӧ���������������л����һ�����
����⣺Fԭ���������ӣ�������H��G����������븺������ȣ����������������Ǵ����Ķ���������G��C��HԪ�ص�����ɫ��Ӧ�ʻ�ɫ��ӦΪNa��A��Mg��B��Al��C��S��D��Cl��E��O��
��1��Al�����ڱ���λ�ڵ������ڵڢ�A�壬�ʴ�Ϊ���������ڵڢ�A�壻
��2��ͬ���ڴ���������������Ӧˮ�������������ǿ��ͬ������ϵ�������������Ӧˮ���������������������������ǿ�������Ͻǣ�����Fû�����ۣ�������HClO4���ʴ�Ϊ��HClO4��
��3���Ȼ�þ��þ���Ӻ�������֮��ͨ�����Ӽ��γɵ����ӻ�������γɹ��̱�ʾΪ�� ���ʴ�Ϊ��
���ʴ�Ϊ�� ��
��
��4��H��E�γ�ԭ�Ӹ�����Ϊ1��1�Ļ�����Ϊ�������ƣ����������ǷǼ��Թ��ۼ��������Ӻ�����֮�������Ӽ����ʴ�Ϊ��Na2O2���Ӽ��ͷǼ��Լ���
��5��F��G�γɵ���Ļ������Ǽ��飬���ĵ���ʽΪ��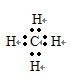 ���ռ�ṹ�����������ͣ�
���ռ�ṹ�����������ͣ�
�ʴ�Ϊ��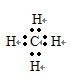 ���������壻
���������壻
��6��ˮ�ķе��������ķе㣬��Ϊˮ���Ӽ����������ʴ�Ϊ��ˮ���Ӽ���������
��7������������ڹ��յ����������ɵ������л������������ˮ�ģ�����״Һ�Σ��Ȼ����Ǽ�������ˮ�����壬����ˮ�γ�����Һ�Σ������ְ������ʴ�Ϊ���Թܱڳ�����״Һ�Σ��Թ���������������
��1��Al�����ڱ���λ�ڵ������ڵڢ�A�壬�ʴ�Ϊ���������ڵڢ�A�壻
��2��ͬ���ڴ���������������Ӧˮ�������������ǿ��ͬ������ϵ�������������Ӧˮ���������������������������ǿ�������Ͻǣ�����Fû�����ۣ�������HClO4���ʴ�Ϊ��HClO4��
��3���Ȼ�þ��þ���Ӻ�������֮��ͨ�����Ӽ��γɵ����ӻ�������γɹ��̱�ʾΪ��
 ���ʴ�Ϊ��
���ʴ�Ϊ�� ��
����4��H��E�γ�ԭ�Ӹ�����Ϊ1��1�Ļ�����Ϊ�������ƣ����������ǷǼ��Թ��ۼ��������Ӻ�����֮�������Ӽ����ʴ�Ϊ��Na2O2���Ӽ��ͷǼ��Լ���
��5��F��G�γɵ���Ļ������Ǽ��飬���ĵ���ʽΪ��
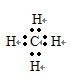 ���ռ�ṹ�����������ͣ�
���ռ�ṹ�����������ͣ��ʴ�Ϊ��
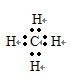 ���������壻
���������壻��6��ˮ�ķе��������ķе㣬��Ϊˮ���Ӽ����������ʴ�Ϊ��ˮ���Ӽ���������
��7������������ڹ��յ����������ɵ������л������������ˮ�ģ�����״Һ�Σ��Ȼ����Ǽ�������ˮ�����壬����ˮ�γ�����Һ�Σ������ְ������ʴ�Ϊ���Թܱڳ�����״Һ�Σ��Թ���������������
������������Ԫ�����ڱ���Ҫ������Ԫ���Լ�����������ʣ��������ݱȽϼ�

��ϰ��ϵ�д�
�����Ŀ
Ԫ�������ɺ�Ԫ�����ڱ���ѧϰ��ѧ����Ҫ���ߣ�������������ȷ���ǣ�������
| A�������ң�Ԫ�����ڱ��еĵ�ʮ����ΪVA�� | B��ijIIA��Ԫ�ص�ԭ������Ϊx��������ͬ���ڵ�IIIA��Ԫ�ص�ԭ����������Ϊx+25 | C��VIA��Ԫ�أ���ԭ�Ӱ뾶����Ӧ��̬�⻯����ȶ�����ǿ | D��53��Ԫ��λ�����ڱ��е�5����VIIA�� |
 NH4++OH-
NH4++OH-