��Ŀ����
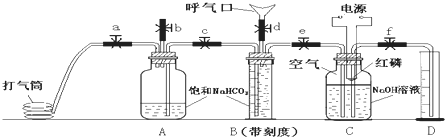
ijѧϰС����������װ��̽����������֮��ķ�Ӧ������A��F�ֱ�Ϊ�����Ͱ����ķ���װ�ã�DΪ�����������Ͱ����ķ�Ӧװ�á�
��ش��������⣺
(1) װ��F����ƿ�ڵĹ����ѡ��_____________��
A���Ȼ��� B����ʯ�� C������������ D���ռ�
(2)ʵ�������õ�Ũ�������������Ϊ36.5%�����ܶ�Ϊ1.19 g/cm3,�������ʵ���Ũ��Ϊ__________(����һλС��)���ڱ������____________L(����һλС��)�Ȼ�������1Lˮ�вſɵô�Ũ�ȵ����ᡣ
(3)���߿���Ӧ���ӱ�Ҫ�ij���װ�ã��뽫����װ������Ӧ���Լ������������пո��У�B__________��C_____________��E_____________��
(4)װ��D�ڳ��ְ��̲��������ڱ����ᣬ�����ù����е�����������Ҫ���Լ���: __________��д��װ��D�з�Ӧ�Ļ�ѧ����ʽ___________���÷�Ӧ���������뻹ԭ�����ʵ���֮��Ϊ____________��
(1) װ��F����ƿ�ڵĹ����ѡ��_____________��
A���Ȼ��� B����ʯ�� C������������ D���ռ�
(2)ʵ�������õ�Ũ�������������Ϊ36.5%�����ܶ�Ϊ1.19 g/cm3,�������ʵ���Ũ��Ϊ__________(����һλС��)���ڱ������____________L(����һλС��)�Ȼ�������1Lˮ�вſɵô�Ũ�ȵ����ᡣ
(3)���߿���Ӧ���ӱ�Ҫ�ij���װ�ã��뽫����װ������Ӧ���Լ������������пո��У�B__________��C_____________��E_____________��
(4)װ��D�ڳ��ְ��̲��������ڱ����ᣬ�����ù����е�����������Ҫ���Լ���: __________��д��װ��D�з�Ӧ�Ļ�ѧ����ʽ___________���÷�Ӧ���������뻹ԭ�����ʵ���֮��Ϊ____________��
(1)BD
(2)11.9mol/L��352.7
(3)����ʳ��ˮ��Ũ�����ʯ��
(4)NaOH��Һ��ʪ��ĺ�ɫʯ����ֽ ��8NH3+3Cl2 = N2+6NH4Cl ��3��2
(2)11.9mol/L��352.7
(3)����ʳ��ˮ��Ũ�����ʯ��
(4)NaOH��Һ��ʪ��ĺ�ɫʯ����ֽ ��8NH3+3Cl2 = N2+6NH4Cl ��3��2

��ϰ��ϵ�д�
 A�ӽ��� ϵ�д�
A�ӽ��� ϵ�д� ȫ�Ų��Ծ�ϵ�д�
ȫ�Ų��Ծ�ϵ�д�
�����Ŀ
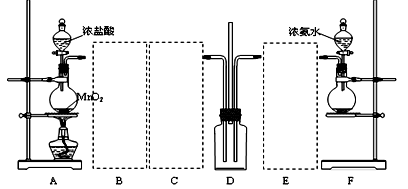
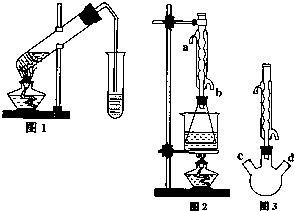 ������������Ҫ�Ĺ�ҵԭ�Ϻ��ܼ���ͨ�����������Ҵ������ᣮijѧϰС������ͼ1װ����ȡ���������ֲ�Ʒ���ٷ������������ĺ�����
������������Ҫ�Ĺ�ҵԭ�Ϻ��ܼ���ͨ�����������Ҵ������ᣮijѧϰС������ͼ1װ����ȡ���������ֲ�Ʒ���ٷ������������ĺ�����| �������� | �Ҵ� | ���� | |
| �е� | 77.1�� | 78.5�� | 117.9�� |
��I��ȷ����20.0g����������Ʒ����ƿ�У���0.50mol?L-1NaOH�ζ�����̪��ָʾ�������յ�ʱ����NaOH��Һ�����Ϊ40.0mL
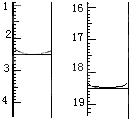
��II����ȡ20.0g���������ֲ�Ʒ��250mL��ƿ�У�����100mL 2.1mol?L-1NaOH��Һ��Ͼ��Ⱥ�װ�������䣬��ˮԡ�ϼ��Ȼ���Լ1Сʱ��װ����ͼ2��ʾ������ȴ����0.50mol?L-1HCl�ζ�������NaOH���յ�ʱ������������Ϊ20.0mL��
�ش��������⣺
��1��ʵ�飨II������ˮ����ˮ���ܵ�
��2������ʵ�飨I������II�����������ݼ���ֲ�����������������������Ϊ
��3��ʵ�������ͬѧ�ǶԴֲ�Ʒ�����������ĺ������߽������ۣ�
��������Ϊ��ʵ�飨II�������������齫ͼ2�е���ƿ��Ϊ����ƿ��װ����ͼ3��������ƿ��c��d��װ����ص�����������ǡ���IJ�����������߲ⶨ�ľ�ȷ�ȣ�����Ϊ������ƿ��c��d��װ����ص�����������ǣ�
A��װ���¶ȼƣ��ϸ���Ʒ�Ӧ�¶�
B��ʵ���о�����ƿ�ڣ��ò������н���
C���ڷ�Ӧ���ڣ������Ȱ�װ�ķ�Һ©������һ������NaOH��Һ
�ڻ���ͬѧ��Ϊ�Ľ�������������ȡװ�ã�ͼ1��������߲��ʣ�������һ���Ľ�����