��Ŀ����
�±�ΪԪ�����ڱ���һ���֣������Ԫ�آ١����ڱ��е�λ�ã�����Ӧ��ѧ����ش��������⣺
| ������ | IA | | 0 | |||||
| 1 | �� | ��A | ��A | ��A | ��A | ��A | ��A | |
| 2 | | | | �� | �� | �� | | |
| 3 | �� | | �� | �� | | �� | �� | |
��2���ܢݢ��γɵļ����Ӱ뾶�ɴ�С��˳��Ϊ_______________�������ӷ��ţ����ۢߢ������������Ӧˮ�����������ǿ������˳��Ϊ____���ѧʽ����
��3���ݺ͢��γɻ�����ľ�������Ϊ__________��
��4����ЩԪ���γɵ��������У�������ˮ����������ǿ��������ǿ�Ӧ����___________���ѧʽ����д������ݵ�����������Ӧˮ�������Ӧ�����ӷ���ʽ__________��
��5��X��Y�ɢ٢ڢ��е����ֻ�����Ԫ����ɡ�X����Һ����С�մ�Ӧ����Y����X��������ϵ������ķ��ӣ�����Է�������Ϊ46����X������Ϊ ��д��X��Һ��С�մ�Ӧ�Ļ�ѧ����ʽΪ_______________��
��1�� ��1�֣���
��1�֣��� ��1�֣���
��1�֣��� ��1�֣���
��1�֣���
��2��S2->O2->Na+(1��)��HClO4>HNO3>H2SiO3(1��) ��3�����Ӿ���(1��)
��4��Al2O3(1��) ��Al2O3+2OH����2AlO�� 2+H2O(2��)
��5������(1��)��HCOOH+NaHCO3�� HCOONa +CO2��+H2O(2��)
�����������������Ԫ�آ١����ڱ��е�λ�ÿ�֪�����Ƿֱ���H��C��N��O��Na��Al��Si��S��Cl��
��1���γɻ������������Ԫ����̼Ԫ�أ�̼Ԫ�ص�ԭ��������6����ԭ�ӽṹʾ��ͼΪ ��ԭ�Ӱ뾶��С������Ԫ�أ��������ԭ�Ӱ뾶��С��ԭ���γ�10������Ϊ��������ṹ�Ļ������Ǽ��飬�����ʽΪ
��ԭ�Ӱ뾶��С������Ԫ�أ��������ԭ�Ӱ뾶��С��ԭ���γ�10������Ϊ��������ṹ�Ļ������Ǽ��飬�����ʽΪ ���ݺ͢��γɻ����������ƣ��������Ӽ������ӻ�������γɹ��̿ɱ�ʾΪ
���ݺ͢��γɻ����������ƣ��������Ӽ������ӻ�������γɹ��̿ɱ�ʾΪ ��
��
��2���ܢݢ��γɵļ����ӷֱ���O2����Na����S2�������ӵĺ�����Ӳ���Խ�����Ӱ뾶Խ��������Ų���ͬ���������Ӱ뾶��ԭ���������������С�����Ԣܢݢ��γɵļ����Ӱ뾶�ɴ�С��˳����S2->O2->Na+���ǽ�����Խǿ������������Ӧˮ���������Խǿ���ۢߢ�����Ԫ�صķǽ�����ǿ��˳����Cl��N��Si��������������Ӧˮ�����������ǿ������˳��ΪHClO4��HNO3��H2SiO3��
��3���ݺ͢��γɻ�������NaCl���侧������Ϊ���Ӿ��塣
��4��������ˮ����������ǿ��������ǿ�Ӧ����Al2O3���Ƶ�����������Ӧˮ�������������ƣ�����������Ӧ�����ӷ���ʽΪAl2O3+2OH����2AlO�� 2+H2O��
��5��X����Һ����С�մ�Ӧ����Y����˵��X������ǿ��̼��ġ�����ΪX����H��C��OԪ��ɣ����X��������ϵ������ķ��ӣ�����Է�������Ϊ46����Xһ���Ǽ��ᣬ��̼�����ɷ�Ӧ�Ļ�ѧ����ʽΪHCOOH+NaHCO3�� HCOONa +CO2��+H2O��
���㣺����Ԫ�����ڱ��Ľṹ�Լ�Ԫ�������ɵ�Ӧ��

�±���Ԫ�����ڱ���һ���֣�����������ĸ�ֱ����ijһ��ѧԪ�ء��û�ѧ����ش��������⡣
| a | | | |||||||||||||||
| | | | | | | | | | | | | b | c | d | | | |
| e | | f | g | | h | i | | ||||||||||
| | | | | | | | | | | | | | | | | | |
| | | | | | | | | | | | | | | | | | |
��1��d��e��f�����Ӱ뾶�ɴ�С��˳��Ϊ
b��c��g����ۺ������������ǿ������˳��Ϊ
a��g��h��i�γɵļ�������ȶ�����ǿ������˳��Ϊ
��2��iԪ�ص�����������Ӧ��ˮ����Ϊ ������e������������Ӧˮ���ﷴӦ�����ӷ���ʽΪ ��
ʵ������ȡi���ʵĻ�ѧ����ʽΪ
��3�������Ե�������������ǿ������ӷ���ʽΪ
��4��e��d���γ�һ�ֵ���ɫ�Ļ��������Ļ�ѧ������Ϊ ����h�ĵͼ�̬�����ﷴӦ�Ļ�ѧ����ʽΪ ��
���ݱ�����Ϣ�ش��������⡣
| Ԫ�� | Si | P | S | Cl |
| ���������� ��Ӧ������ | ���� | �������������ܷ�Ӧ | ���� | ���ջ��ȼʱ������ը������ |
��1��S��Ԫ�����ڱ��е�λ���� ��
��2�����ݱ�����Ϣ��֪��Si��P��S��Cl ����Ԫ�صĵķǽ�����������ǿ����ԭ�ӽṹ����ԭ��ͬ����Ԫ�ص��Ӳ�����ͬ���������ң� ��ԭ�Ӱ뾶��С���õ�����������ǿ��
��3��25��ʱ����������Ԫ�صĵ�����������Ӧ����l mol��̬�⻯��ķ�Ӧ�����£�
a��+34.3 kJ��mol-1 b��+9.3 kJ��mol-1 c��?20.6 kJ��mol-1 d��?92.3 kJ��mol-1
��д����̬���ף�P4����H2��Ӧ������̬�⻯����Ȼ�ѧ����ʽ ��
��4��̽��ͬ����Ԫ�����ʵ�һЩ��ͬ���ɣ���ѧϰ��ѧ����Ҫ����֮һ����֪����Se��������������Ԫ�أ��䲿����Ϣ��ͼ��
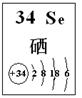
�������й�˵����ȷ���� ������ĸ����
a. ԭ�Ӱ뾶��Se��S��P b. �ȶ��ԣ�H2Se��H2S
c. ��Ϊ����H2Se��HCl�����Էǽ�����Se��Cl
d. SeO2����������������ռ���Һ��Ӧ
�����±����г���H2SeO3���ֲ�ͬ��ѧ���ʵ��Ʋ⣬������д����Ӧ�Ļ�ѧ����ʽ��
| ��� | �����Ʋ� | ��ѧ����ʽ |
| 1 | ������ | H2SeO3��4HI=Se����2I2��3H2O |
| 2 | | |
| 3 | | |
�±���Ԫ�����ڱ���һ���֣����е�ÿ����ĸ��ʾһ�ֶ�����Ԫ��,�ش��������⣺
| | | | ||||||
| | | | | | | A | B | |
| C | | | D | E | F | G] | | |
��1������G��ԭ�ӽṹʾ��ͼ___________________
��2��A��B���á�E��F����Ԫ�ص�ԭ�Ӱ뾶������___________���ѧʽ��
��3��E��F��GԪ�ص�����������Ӧˮ��������Խ�������_____________���û�ѧʽ��ʾ��
��4��д��C�ͣ��γɻ�����ĵ���ʽ______________________
�±��г���A��G7��Ԫ�������ڱ��е�λ�ã�
| �� ���� | ��A | ��A | ��A | ��A | ��A | ��A | ��A | 0 |
| 2 | | | | C | D | E | | |
| 3 | | | B | | | | F | G |
| 4 | A | | | | | | | |
��1���ֱ�д��A��D��G��Ԫ�ط��ţ� �� �� �����л�ѧ��������õ��� ����Ԫ�ط��ţ���
��2��F���⻯���л�ѧ���������� ��F������������Ӧ��ˮ����Ļ�ѧʽ�� ��
��3��C��D��E����Ԫ���У��⻯������������ ��
��4��д��B��F�γɵĻ������ˮ�ⷴӦ�����ӷ���ʽ ��
