��Ŀ����
��Ҫ�Ļ���ԭ��M��C5H8O4�����������ζ������������ͼ��ʾ�������̺ϳɣ�
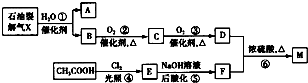
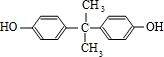
��֪��A��B��Ϊͬ���칹�壻X���ܶ�����ͬ������H2�ܶȵ�21����D��F��M������NaHCO3?��Һ��Ӧ�������壮
��1��A��ϵͳ������������Ϊ
��2��X�Ľṹ��ʽ��
��3���١��ķ�Ӧ������ȡ����Ӧ����
��4��д������ʽ��B��C
��5��M��ͬ���칹���кܶ࣬����һ��ͬ���칹��ֻ����һ�ֹ����ţ������Ժͼ��������¶���ˮ�����������л����ͬ���칹��Ľṹ��ʽ��

��֪��A��B��Ϊͬ���칹�壻X���ܶ�����ͬ������H2�ܶȵ�21����D��F��M������NaHCO3?��Һ��Ӧ�������壮
��1��A��ϵͳ������������Ϊ
2-����
2-����
����2��X�Ľṹ��ʽ��
CH2=CHCH3
CH2=CHCH3
��E�Ľṹ��ʽ��ClCH2COOH
ClCH2COOH
����3���١��ķ�Ӧ������ȡ����Ӧ����
�ܢݢ�
�ܢݢ�
����4��д������ʽ��B��C
2CH3CH2CH2OH+O2
2CH3CH2CHO+2H2O
| ���� |
| �� |
2CH3CH2CH2OH+O2
2CH3CH2CHO+2H2O
��D+F��M| ���� |
| �� |
CH3CH2COOH+HOCH2COOH
CH3CH2COOCH2COOH+H2O
| Ũ���� |
| �� |
CH3CH2COOH+HOCH2COOH
CH3CH2COOCH2COOH+H2O
��| Ũ���� |
| �� |
��5��M��ͬ���칹���кܶ࣬����һ��ͬ���칹��ֻ����һ�ֹ����ţ������Ժͼ��������¶���ˮ�����������л����ͬ���칹��Ľṹ��ʽ��
CH3OOCCH2COOCH3��HCOOCH2CH2CH2OOCH��CH3COOCH2OOCCH3
CH3OOCCH2COOCH3��HCOOCH2CH2CH2OOCH��CH3COOCH2OOCCH3
��������X��ʯ���ѽ�������������X���ܶ�����ͬ������H2�ܶȵ�21������X����Է�������Ϊ21��2=42����XΪCH2=CHCH3�����Ƿ����ӳɷ�Ӧ������A��B��A��B��Ϊͬ���칹�壬�ʶ���Ϊ����B��������C��C��������D����BΪCH3CH2CH2OH��AΪCH3CH��OH��CH3��D����NaHCO3��Һ��Ӧ�������壬��D�����Ȼ���˳�ƿ�֪��CΪCH3CH2CHO��DΪCH3CH2COOH��F��M������NaHCO3��Һ��Ӧ�������壬�ʶ������Ȼ���CH3COOH�������ڹ�������������E��EΪClCH2COOH������������ˮ��Һ��ˮ�⡢Ȼ���ữ�õ�F����FΪHOCH2COOH����CH3CH2COOH������Ӧ����M��M����������ζ��Ӧ����������Ӧ����MΪCH3CH2COOCH2COOH���ݴ˽��
����⣺X��ʯ���ѽ�������������X���ܶ�����ͬ������H2�ܶȵ�21������X����Է�������Ϊ21��2=42����XΪCH2=CHCH3�����Ƿ����ӳɷ�Ӧ������A��B��A��B��Ϊͬ���칹�壬�ʶ���Ϊ����B��������C��C��������D����BΪCH3CH2CH2OH��AΪCH3CH��OH��CH3��D����NaHCO3��Һ��Ӧ�������壬��D�����Ȼ���˳�ƿ�֪��CΪCH3CH2CHO��DΪCH3CH2COOH��F��M������NaHCO3��Һ��Ӧ�������壬�ʶ������Ȼ���CH3COOH�������ڹ�������������E��EΪClCH2COOH������������ˮ��Һ��ˮ�⡢Ȼ���ữ�õ�F����FΪHOCH2COOH����CH3CH2COOH������Ӧ����M��M����������ζ��Ӧ����������Ӧ����MΪCH3CH2COOCH2COOH��
��1��������������֪��AΪCH3CH��OH��CH3��ϵͳ����������Ϊ��2-�������ʴ�Ϊ��2-������
��2��������������֪��X�Ľṹ��ʽ��CH2=CHCH3��E�Ľṹ��ʽΪ��ClCH2COOH���ʴ�Ϊ��CH2=CHCH3��ClCH2COOH��
��3����Ϊ�ӳɷ�Ӧ���ڢ�Ϊ������Ӧ���ܢݢ�����ȡ����Ӧ���ʴ�Ϊ���ܢݢޣ�
��4��B��C�ķ�Ӧ����ʽΪ��2CH3CH2CH2OH+O2
2CH3CH2CHO+2H2O��
D+F��M�ķ�Ӧ����ʽΪ��CH3CH2COOH+HOCH2COOH
CH3CH2COOCH2COOH+H2O��
�ʴ�Ϊ��2CH3CH2CH2OH+O2
2CH3CH2CHO+2H2O��CH3CH2COOH+HOCH2COOH
CH3CH2COOCH2COOH+H2O��
��5��M��ͬ���칹���кܶ࣬����һ��ͬ���칹��ֻ����һ�ֹ����ţ������Ժͼ��������¶���ˮ�����������л�����ͬ���칹�庬��2����������ͬ���칹��Ľṹ��ʽ�ǣ�CH3OOCCH2COOCH3��HCOOCH2CH2CH2OOCH��CH3COOCH2OOCCH3��
�ʴ�Ϊ��CH3OOCCH2COOCH3��HCOOCH2CH2CH2OOCH��CH3COOCH2OOCCH3��
��1��������������֪��AΪCH3CH��OH��CH3��ϵͳ����������Ϊ��2-�������ʴ�Ϊ��2-������
��2��������������֪��X�Ľṹ��ʽ��CH2=CHCH3��E�Ľṹ��ʽΪ��ClCH2COOH���ʴ�Ϊ��CH2=CHCH3��ClCH2COOH��
��3����Ϊ�ӳɷ�Ӧ���ڢ�Ϊ������Ӧ���ܢݢ�����ȡ����Ӧ���ʴ�Ϊ���ܢݢޣ�
��4��B��C�ķ�Ӧ����ʽΪ��2CH3CH2CH2OH+O2
| ���� |
| �� |
D+F��M�ķ�Ӧ����ʽΪ��CH3CH2COOH+HOCH2COOH
| Ũ���� |
| �� |
�ʴ�Ϊ��2CH3CH2CH2OH+O2
| ���� |
| �� |
| Ũ���� |
| �� |
��5��M��ͬ���칹���кܶ࣬����һ��ͬ���칹��ֻ����һ�ֹ����ţ������Ժͼ��������¶���ˮ�����������л�����ͬ���칹�庬��2����������ͬ���칹��Ľṹ��ʽ�ǣ�CH3OOCCH2COOCH3��HCOOCH2CH2CH2OOCH��CH3COOCH2OOCCH3��
�ʴ�Ϊ��CH3OOCCH2COOCH3��HCOOCH2CH2CH2OOCH��CH3COOCH2OOCCH3��
���������⿼���л�����ƶ���ϳɣ��Ѷ��еȣ�ȷ��X������ǽ���ؼ�����Ϸ�Ӧ������������Ӧ������˳�Ʒ��ƶϣ�ע�����չ����ŵ����ʣ�

��ϰ��ϵ�д�
 ȫ�ŵ�����Ԫ�ƻ�ϵ�д�
ȫ�ŵ�����Ԫ�ƻ�ϵ�д�
�����Ŀ



 �Ļ�ѧ����ʽ��
�Ļ�ѧ����ʽ��


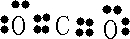
 H++Cl-+HClO
H++Cl-+HClO
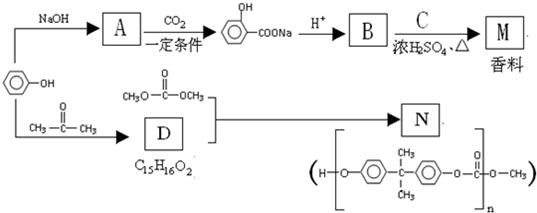
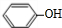 +NaOH��
+NaOH��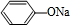 +H2O
+H2O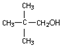
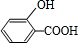 +
+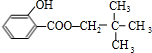 +H2O
+H2O