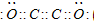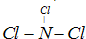��Ŀ����
����A��B��C��D��E��F��G���ֺ˵����С��18��Ԫ�أ����ǵ�ԭ������һ�ε��������У�Aԭ�Ӻ��������ӣ�Bԭ�������������Ǵ�����2����Dԭ������������������Ӳ�����3����Eԭ�Ӻ����һ��ȵ������������1��F���������������ڵ�һ���������G�γɵ�-1��������Arԭ�Ӻ�������Ų���ͬ��
�ɴ���֪��
��1��Aԭ�ӷ�����
��2��Bԭ�ӽṹʾ��ͼ��

��3��GԪ���γɵĵ�����A��D��E��Ԫ���γɳʼ��ԵĻ����ﷴӦ�����ӷ���ʽ��
����F�ֱ���C���ʡ�BDԪ���γɵĻ�������ȼ�յĻ�ѧ����ʽ��
�ɴ���֪��
��1��Aԭ�ӷ�����
11H
11H
����2��Bԭ�ӽṹʾ��ͼ��


��3��GԪ���γɵĵ�����A��D��E��Ԫ���γɳʼ��ԵĻ����ﷴӦ�����ӷ���ʽ��
2OH-+Cl2�TCl-+ClO-+H2O
2OH-+Cl2�TCl-+ClO-+H2O
����F�ֱ���C���ʡ�BDԪ���γɵĻ�������ȼ�յĻ�ѧ����ʽ��
3Mg+N2
Mg3N2
| ||
3Mg+N2
Mg3N2
��
| ||
2Mg+CO2
2MgO+C
| ||
2Mg+CO2
2MgO+C
��
| ||
������A��B��C��D��E��F��G���ֺ˵����С��18��Ԫ�أ����ǵ�ԭ���������ε��������У�Aԭ�Ӻ��������ӣ���A��HԪ�أ�Bԭ�������������Ǵ�����2������B��CԪ�أ�Dԭ������������������Ӳ�����3������D��OԪ�أ�C��ԭ����������BС��D������C��NԪ�أ�Eԭ�Ӻ����һ��ȵ������������1����E��NaԪ�أ�F���������������ڵ�һ�����������F��ԭ����������E������F��MgԪ�أ�G�γɵ�-1��������Arԭ�Ӻ�������Ų���ͬ����G��ClԪ�أ�
����⣺A��B��C��D��E��F��G���ֺ˵����С��18��Ԫ�أ����ǵ�ԭ���������ε��������У�Aԭ�Ӻ��������ӣ���A��HԪ�أ�Bԭ�������������Ǵ�����2������B��CԪ�أ�Dԭ������������������Ӳ�����3������D��OԪ�أ�C��ԭ����������BС��D������C��NԪ�أ�Eԭ�Ӻ����һ��ȵ������������1����E��NaԪ�أ�F���������������ڵ�һ�����������F��ԭ����������E������F��MgԪ�أ�G�γɵ�-1��������Arԭ�Ӻ�������Ų���ͬ����G��ClԪ�أ�
��1����AΪHԪ�أ���Aԭ�ӷ�����11H���ʴ�Ϊ��11H��
��2��BΪCԪ�أ�Cԭ�ӽṹʾ��ͼΪ ���ʴ�Ϊ��
���ʴ�Ϊ�� ��
��
��3��GԪ���γɵĵ�����������A��D��E��Ԫ���γɳʼ��ԵĻ��������������ƣ�����������������Һ��Ӧ�����ӷ���ʽΪ��2OH-+Cl2�TCl-+ClO-+H2O��þ�ֱ��ڵ�����������̼��ȼ�յĻ�ѧ����ʽΪ��3Mg+N2
Mg3N2��2Mg+CO2
2MgO+C���ʴ�Ϊ��2OH-+Cl2�TCl-+ClO-+H2O��3Mg+N2
Mg3N2��2Mg+CO2
2MgO+C��
��1����AΪHԪ�أ���Aԭ�ӷ�����11H���ʴ�Ϊ��11H��
��2��BΪCԪ�أ�Cԭ�ӽṹʾ��ͼΪ
 ���ʴ�Ϊ��
���ʴ�Ϊ�� ��
����3��GԪ���γɵĵ�����������A��D��E��Ԫ���γɳʼ��ԵĻ��������������ƣ�����������������Һ��Ӧ�����ӷ���ʽΪ��2OH-+Cl2�TCl-+ClO-+H2O��þ�ֱ��ڵ�����������̼��ȼ�յĻ�ѧ����ʽΪ��3Mg+N2
| ||
| ||
| ||
| ||
���������⿼��ԭ�ӽṹ�����ʵĹ�ϵ����ȷ�ƶ�Ԫ���ǽⱾ��Ĺؼ���ע��þ�ڶ�����̼����ȼ�գ�Ϊ�״��㣮

��ϰ��ϵ�д�
 ��У����ϵ�д�
��У����ϵ�д�
�����Ŀ
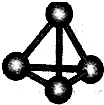 ����A��B��C��D��E��F���ֶ�����Ԫ�أ����ǵ�ԭ��������������D��E���⻯����ӹ��Ͷ���V�ͣ�A��B������������֮����C��������������ȣ�A�ֱܷ���B��C��D�γɵ���������ȵķ��ӣ���A��D���γɵĻ���������¾�ΪҺ̬��
����A��B��C��D��E��F���ֶ�����Ԫ�أ����ǵ�ԭ��������������D��E���⻯����ӹ��Ͷ���V�ͣ�A��B������������֮����C��������������ȣ�A�ֱܷ���B��C��D�γɵ���������ȵķ��ӣ���A��D���γɵĻ���������¾�ΪҺ̬��