��Ŀ����
�����Ԫ�����ڱ���һ���֣��г���ʮ��Ԫ�������ڱ��е�λ�ã�
���û�ѧ����ش���������
��1��ʮ��Ԫ���л�ѧ��������õ���
��2��Ԫ�آ���Ԫ�آ��γɵĻ�����ķ���ʽ
��3���ߺ�Ԫ���⻯��ķ���ʽ��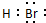
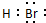 ���ߺ�Ԫ���⻯���Ԫ�آڷ�Ӧ�����ӷ���ʽ��
���ߺ�Ԫ���⻯���Ԫ�آڷ�Ӧ�����ӷ���ʽ��
��4���١��ۺ͢�����Ԫ������������ˮ����ļ�����ǿ����˳�����
��5���ڡ��ۺ͢�����Ԫ�����γɵļ����ӵİ뾶��С�����ǣ�
| �� ���� |
IA | IIA | IIIA | VIA | VA | VIA | VIIA | 0 |
| 2 | �� | �� | ||||||
| 3 | �� | �� | �� | �� | �� | |||
| 4 | �� | �� | �� |
��1��ʮ��Ԫ���л�ѧ��������õ���
Ar
Ar
��Ԫ�آ���Ԫ�آ��γɵĻ�����������
����
����ϣ���Թ��ۼ����Ǽ��Թ��ۼ������Ӽ�������2��Ԫ�آ���Ԫ�آ��γɵĻ�����ķ���ʽ
CCl4
CCl4
��������������
����
�������ۻ������ӣ�����3���ߺ�Ԫ���⻯��ķ���ʽ��
H2O
H2O
�����Ԫ���⻯��ĵ���ʽ��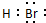
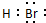
2K+2H2O�T2K++2OH-+H2��
2K+2H2O�T2K++2OH-+H2��
����4���١��ۺ͢�����Ԫ������������ˮ����ļ�����ǿ����˳�����
NaOH
NaOH
��Mg��OH��2
Mg��OH��2
��Al��OH��3
Al��OH��3
����5���ڡ��ۺ͢�����Ԫ�����γɵļ����ӵİ뾶��С�����ǣ�
Mg2+
Mg2+
��Ca2+
Ca2+
��K+
K+
������������Ԫ�����ڱ���֪����ΪNa����ΪK����ΪMg����ΪCa����ΪAl����ΪC����ΪO����ΪCl����ΪBr����ΪAr������Ԫ�صĵ��ʵ����ʼ�������Խǿ����Ӧ������������ˮ����ļ���Խǿ���Ƚϰ뾶�ȿ��㣬������ͬ�Ų������Ӱ뾶ԽС����ԭ������Խ���Դ������
����⣺��Ԫ�����ڱ���֪����ΪNa����ΪK����ΪMg����ΪCa����ΪAl����ΪC����ΪO����ΪCl����ΪBr����ΪAr��
��1������Ԫ���У�ֻ��Ar������������Ϊ8��Ϊ�ȶ��ṹ����ѧ��������ã�Na��Br�γ����ӻ�����NaBr�������Ӽ���ϣ��ʴ�Ϊ��Ar�����ӣ�
��2��Cl��C�γɵĻ�����ΪCCl4���Թ��ۼ���ϣ������ڹ��ۻ�����ʴ�Ϊ��CCl4�����ۣ�
��3����Ԫ�ص��⻯��ΪH2O��Br���⻯��ΪHBr��Ϊ���ۻ���������ʽΪ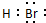 ��ˮ��ط�Ӧ�����������غ��������仯ѧ��Ӧ����ʽΪ2K+2H2O�T2KOH+H2�������ӷ�ӦΪ2K+2H2O�T2K++2OH-+H2����
��ˮ��ط�Ӧ�����������غ��������仯ѧ��Ӧ����ʽΪ2K+2H2O�T2KOH+H2�������ӷ�ӦΪ2K+2H2O�T2K++2OH-+H2����
�ʴ�Ϊ��H2O��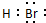 ��2K+2H2O�T2K++2OH-+H2����
��2K+2H2O�T2K++2OH-+H2����
��4��Na��Mg��Al��ͬ���ڣ�������ΪNa��Mg��Al������Ԫ������������ˮ����ļ�����ǿ����˳�����
NaOH��Mg��OH��2��Al��OH��3��
�ʴ�Ϊ��NaOH��Mg��OH��2��Al��OH��3��
��5����ΪK����ΪMg����ΪCa��þ������2�����Ӳ㣬�����Ӱ뾶��С����������������Ӿ�����ͬ�ĵ��Ӳ�ṹ�����Ӱ뾶ԽС����ԭ������Խ��
�����Ӱ뾶�Ĺ�ϵΪMg2+��Ca2+��K+���ʴ�Ϊ��Mg2+��Ca2+��K+��
��1������Ԫ���У�ֻ��Ar������������Ϊ8��Ϊ�ȶ��ṹ����ѧ��������ã�Na��Br�γ����ӻ�����NaBr�������Ӽ���ϣ��ʴ�Ϊ��Ar�����ӣ�
��2��Cl��C�γɵĻ�����ΪCCl4���Թ��ۼ���ϣ������ڹ��ۻ�����ʴ�Ϊ��CCl4�����ۣ�
��3����Ԫ�ص��⻯��ΪH2O��Br���⻯��ΪHBr��Ϊ���ۻ���������ʽΪ
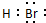 ��ˮ��ط�Ӧ�����������غ��������仯ѧ��Ӧ����ʽΪ2K+2H2O�T2KOH+H2�������ӷ�ӦΪ2K+2H2O�T2K++2OH-+H2����
��ˮ��ط�Ӧ�����������غ��������仯ѧ��Ӧ����ʽΪ2K+2H2O�T2KOH+H2�������ӷ�ӦΪ2K+2H2O�T2K++2OH-+H2�����ʴ�Ϊ��H2O��
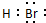 ��2K+2H2O�T2K++2OH-+H2����
��2K+2H2O�T2K++2OH-+H2������4��Na��Mg��Al��ͬ���ڣ�������ΪNa��Mg��Al������Ԫ������������ˮ����ļ�����ǿ����˳�����
NaOH��Mg��OH��2��Al��OH��3��
�ʴ�Ϊ��NaOH��Mg��OH��2��Al��OH��3��
��5����ΪK����ΪMg����ΪCa��þ������2�����Ӳ㣬�����Ӱ뾶��С����������������Ӿ�����ͬ�ĵ��Ӳ�ṹ�����Ӱ뾶ԽС����ԭ������Խ��
�����Ӱ뾶�Ĺ�ϵΪMg2+��Ca2+��K+���ʴ�Ϊ��Mg2+��Ca2+��K+��
���������⿼��λ�á��ṹ�����ʵĹ�ϵ������Ԫ�����ڱ��ó���Ԫ���ǽ����Ĺؼ���Ȼ������Ԫ�ص����ʡ�ͬ����Ԫ�ص����ʱ仯���ɡ����Ӱ뾶�ıȽϹ�������ɣ�

��ϰ��ϵ�д�
 100�ִ�����ĩ���ϵ�д�
100�ִ�����ĩ���ϵ�д�
�����Ŀ
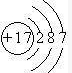

 ���Ŀռ�������Ϊ
���Ŀռ�������Ϊ