��Ŀ����
��15�֣�����������(Na2S2O5)�dz��õ�ʳƷ��������֮һ��ij�о�С���������ʵ�飺
ʵ��һ ���������Ƶ���ȡ
������ͼװ��(ʵ��ǰ�ѳ���װ���ڵĿ���)��ȡNa2S2O5��װ�â�����Na2S2O5���������������ķ�ӦΪ��Na2SO3��SO2��Na2S2O5

��1��װ�â��в�������Ļ�ѧ����ʽΪ ��
��2��Ҫ��װ�â��л���������ľ��壬�ɲ�ȡ�ķ��뷽���� ��
��3��װ�â����ڴ���β������ѡ�õ������װ��(�г���������ȥ)Ϊ (�����)��

ʵ��� ���Ѿ��п��������������IJⶨ
��4�����ѾƳ���Na2S2O5�������������ⶨij���Ѿ��п��������IJ�����(������SO2����)�ķ������£�

(��֪���ζ�ʱ��Ӧ�Ļ�ѧ����ʽΪSO2��I2��2H2O��H2SO4��2HI)
�ٰ���������ʵ�飬���ı�I2��Һ25��00 mL���ô�ʵ������Ʒ�п��������IJ�����(������SO2����)Ϊ g��L��1��
��������ʵ������У����в���HI���������������ý�� (�ƫ�ߡ���ƫ�͡����䡱)��
��1��Na2SO3+H2SO4��Na2SO4+SO2��+H2O��Na2SO3+2H2SO4��2NaHSO4+SO2��+H2O
��2������ ��3��d ��4����0��16 ��ƫ��
��������
�����������1��װ��I������ȡ��������ķ�Ӧ��Ũ�������������Ʒ�Ӧ���ɶ������������ơ�ˮ����ѧ����ʽΪNa2SO3+H2SO4��Na2SO4+SO2��+H2O��Na2SO3+2H2SO4��2NaHSO4+SO2��+H2O��
��2��װ�â�����Na2S2O5�������������Դ�װ��II�еõ�����ķ����ǹ��ˣ�
��3��װ��III����β����β���ijɷ��Ƕ���������������������Һ���գ����ŵ����θ������ɷ�ֹ���������Դ�ѡd��
��4���ٸ���SO2��I2��2H2O��H2SO4��2HI��֪��SO2��I2�����Ը������ĵ�����ʵ����ɼ�����������������0��025L��0��01mol/L��64g/mol=0��016g�����Ըô�ʵ������Ʒ�п��������IJ�������0��016g/0��1L=0��16g/L��
��������ʵ������У����в���HI������������������ĵĵⵥ�ʵ����ʵ������٣����Բⶨ���ƫ�͡�
���㣺�������������ȡ�������������IJⶨʵ�����������㣬����ʵ��������жϣ����ķ���

 ��������һ���þ�ϵ�д�
��������һ���þ�ϵ�д� Сѧ��10����Ӧ����ϵ�д�
Сѧ��10����Ӧ����ϵ�д�ѡ���⡾��ѧһһѡ��ѧ�뼼������15�֣�����п�̳�����Ҫ�ɷ�ΪZnO��������CuO��FeO��Ϊԭ�ϣ�������ȡ�Ȼ�п�ͽ���п��
����ȡ�Ȼ�п��Ҫ�������£�
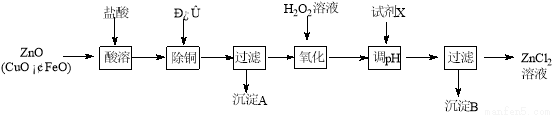
�±��г�����ؽ������������������������pH (��ʼ������pH����������Ũ��Ϊ1.0 mol��L��1����)��
�������� | ��ʼ������pH | ������ȫ��pH |
Fe3+ | 1. 1 | 3. 2 |
Zn2+ | 5. 2 | 6. 4 |
Fe2+ | 5. 8 | 8. 8 |
��1������H2O2��Һ������Ӧ�����ӷ���ʽΪ ��
��2������ͼ�У�Ϊ�˽�����Һ����ȣ��Լ�X�������� ��ѡ����ţ�
a��ZnO��b��Zn(OH)2 ��c��Zn2(OH)2CO3 ��d��ZnSO4��pHӦ������ ��
��3���Ȼ�п�ܴ����ᣨ2���ǻ����ᣩ���ɱ�������C6H8O4���;����ᣬ�������Ľṹ��ʽΪ ��������Ľṹ��ʽΪ ��
����ȡ����п���ü��ܽ�ZnO(s)+2NaOH(aq)+H2O(l)=Na2[Zn(OH)4](aq)��Ȼ�����ȡҺ��
��4����ʯī���缫���ʱ����������������Ϊ �������ĵ缫��ӦΪ ��
��5����п�̳����ü��ܣ������������ܺ��⣬��Ҫԭ���� ��

 2H+ + Cl-+ ClO-
2H+ + Cl-+ ClO-