��Ŀ����
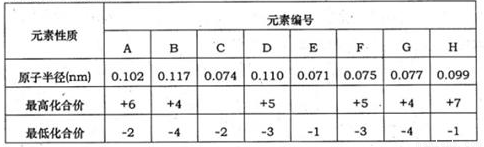
��16�֣��±�Ϊ���ֶ�����Ԫ�ػ��ϼۼ���Ӧ�⻯��е�����ݣ�
|
Ԫ������[ |
Ԫ�ر��[��Դ:ZXXK][��Դ:ZXXK] |
|||||||
|
A |
B |
C |
D |
E |
F |
G |
H |
|
|
�⻯��ķе㣨�棩 |
��60.7 |
��33.4 |
��111.5 |
100 |
��87.7 |
19.54 |
��84.9 |
��161.5 |
|
����ϼ� |
+6 |
+5 |
��4 |
|
+5 |
|
+7 |
��4 |
|
��ͻ��ϼ� |
��2 |
��3 |
��4 |
��2 |
��3 |
��1 |
��1 |
��4 |
��֪��
��A��D���γɻ�����AD2��AD3���������Ʊ�ǿ��ף�
��B��D���γɻ�����BD��BD2���������Ʊ�ǿ���ҡ�
��ش�
��1���������ڵ�������Ԫ�ص��� �� ���ñ���Ԫ�ر����д����
��2��д��H�����������ĵ���ʽ���������������������Ƚ�A��D��G���ּ������ӵİ뾶��С��r(������)��r(������)��r(������) ������ʵ�ʵ�Ԫ�ط��ű�ʾ��
��3���ɱ���DԪ�غ���Ԫ�ص�ԭ�Ӱ�1:1��ɵij���Һ̬�������ϡ��Һ�ױ����ֽ⣬��ʹ�õĴ���Ϊ������ţ�________��_________��
a��MnO2������ b��FeCl3���� c��Na2SO3���� d��KMnO4
��4���������ΪADG2��������ˮ�л�ǿ��ˮ�⣬����ʹƷ����Һ��ɫ����ɫ�����һ��ǿ�ᡣ�÷�Ӧ�Ļ�ѧ����ʽ�ǣ� �� ��
��5����ҵ����ǿ����ʱ���ô�����Һ����β�����÷�Ӧ���£�
BD+BD2+Na2CO3=2 ��������+CO2
���ٺ�����ij�εĻ�ѧʽӦΪ��������������
����ÿ����44.8L����״����CO2��������β���������� �� g��
��6�������һ��ʵ�鷽����ʹ��ͭ��ϡ��ǿ���Ӧ���õ���ɫ��Һ���������ڴ��ָ��λ�û��ʵ��װ��ͼ��������Ҫ��˵�����ɡ�
��1��ACEG��2�֣�
��2��CO2�ĵ���ʽ��2�֣���r(��S2����)��r(��Cl����)��r(��O2����)��2�֣�
��3��a b��2�֣�
��4��SOCl2+H2O��SO2+2HCl��2�֣�
��5����NaNO2��1�֣�����152g����2�֣�
��6�� ��3�֣�
��3�֣�
��������

 Сѧ��ʱ��ҵȫͨ����ϵ�д�
Сѧ��ʱ��ҵȫͨ����ϵ�д� �����ÿ�ʱѵ��ϵ�д�
�����ÿ�ʱѵ��ϵ�д� ��Ԫȫ��������ϵ�д�
��Ԫȫ��������ϵ�д� �»ƸԱ����ܾ�ϵ�д�
�»ƸԱ����ܾ�ϵ�д�

 �±�Ϊ���ֶ�����Ԫ�ػ��ϼۼ�����Ӧԭ�Ӱ뾶�����ݣ���ش��������⣺
�±�Ϊ���ֶ�����Ԫ�ػ��ϼۼ�����Ӧԭ�Ӱ뾶�����ݣ���ش��������⣺