��Ŀ����
�ڲ�ͬ�¶��£�ˮ�ﵽ����ƽ��ʱc��H+����c��OH�����Ĺ�ϵ��ͼ4��ʾ������˵������ȷ����


| A��100��ʱ��pH=12��NaOH��Һ��pH=2��H2SO4��Һǡ���кͣ�������Һ��pH����7 |
| B��25��ʱ,0.2 mol/L Ba(OH)2��Һ��0.2 mol/L HCl��Һ�������ϣ�������Һ��pH����7 |
| C��25��ʱ, 0.2 mol/L NaOH��Һ��0.2 mol/L������Һǡ���кͣ�������Һ��pH����7 |
| D��25��ʱ,pH=12�İ�ˮ��pH=2��H2SO4��Һ�������ϣ�������Һ��pH����7 |
D
���������100��ʱ��pH=12��NaOH��ҺŨ����1mol/L�����Է�Ӧ�����������ǹ����ģ���Һ�Լ��ԣ�A����ȷ��B�����������ǹ����ģ���Һ�Լ��ԣ�B����ȷ��C��ǡ�÷�Ӧ�������ɵĴ�����ˮ�⣬��Һ�Լ��ԣ�C����ȷ��D�а�ˮ��������Һ�Լ��ԣ�D��ȷ����ѡD��
������������Һ��ѡA����������û�п��ǵ�ˮ�����ӻ����������¶ȵ����߶�����ģ������100��ʱ��pH��6ʱ����Һ�������Եġ����������pH�ж���Һ�����ʱ����Ҫע����Һ���¶ȡ�

��ϰ��ϵ�д�
 ���Ӣ��������ϵ�д�
���Ӣ��������ϵ�д�
�����Ŀ
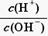 ��1010����ش��������⣺
��1010����ش��������⣺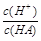
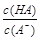
 H����OH���������ƶ�����Һ�ʼ��Ե��ǣ� ��
H����OH���������ƶ�����Һ�ʼ��Ե��ǣ� �� ѡ�õζ���
ѡ�õζ���