��Ŀ����
���и�������ʾ��ͼһ�µ���
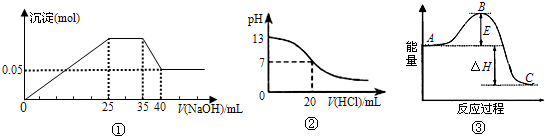
- A.ͼ�ٱ�ʾ��Mg2+��Al3+��NH4+������Һ�еμ�NaOH��Һʱ������������NaOH������Ĺ�ϵͼ�����������ӵ����ʵ���֮��Ϊ��n��Mg2+����n��Al3+����n��NH4+��=2��3��2
- B.ͼ����ʹ�õ�NaOH��Ũ��Ϊ2mol/L
- C.ͼ�ڱ�ʾ25��ʱ����0.1mol?L-1����ζ�20mL0.1mol?L-1NaOH��Һ����Һ��pH�����������ı仯
- D.ͼ�������߱�ʾ3N2��g��+N2��g��
 2NH3��g����Ӧ���̵������仯����ʹ�ô�����B�������
2NH3��g����Ӧ���̵������仯����ʹ�ô�����B�������
C
������ͼ���漰�������ӷ���ʽ�У�Mg2++2OH-�TMg��OH��2����Al3++3OH-�TAl��OH��3����NH4++OH-�TNH3?H2O��
Al��OH��3+OH-�TAlO2-+2H2O����NaOH��Һ��Ũ��Ϊxmol/L����n��NH4+��=0.01xmol��n��Al��OH��3��=n��Al3+��=0.005xmol��
n��Mg2+��= =0.005xmol����n��Mg2+����n��Al3+����n��NH4+��=1��1��2��
=0.005xmol����n��Mg2+����n��Al3+����n��NH4+��=1��1��2��
����������þ�����ʵ�����֪��0.005xmol=0.05mol��x=10mol/L��
ͼ���м����������Ϊ20mlʱ����кͣ�PH=7���кͺ�����������ᣬPH��С����Һ�����ԣ�
ͼ�������߱�ʾ��Ӧ���������������Ĵ�С����A�Ƿ�Ӧ���Ӧ���ȣ���֮���ȣ�B��Ϊ��ܣ������������ܽ��ͣ�
���A��ͼ���漰�������ӷ���ʽ�У�Mg2++2OH-�TMg��OH��2����Al3++3OH-�TAl��OH��3����NH4++OH-�TNH3?H2O��
Al��OH��3+OH-�TAlO2-+2H2O����NaOH��Һ��Ũ��Ϊxmol/L����n��NH4+��=0.01xmol��n��Al��OH��3��=n��Al3+��=0.005xmol��
nn��Mg2+��= =0.005xmol����n��Mg2+����n��Al3+����n��NH4+��=1��1��2����A����
=0.005xmol����n��Mg2+����n��Al3+����n��NH4+��=1��1��2����A����
B����NaOH��Һ��Ũ��Ϊxmol/L������n��Mg2+��=n��Mg��OH��2��=0.005xmol=0.05mol��x=10mol/L����B����
C��ͼ����25��ʱ����0.1mol?L-1����ζ�20mL0.1mol?L-1NaOH��Һ�������������Ϊ20mlʱ����кͣ�PH=7���кͺ�����������ᣬPH��С����Һ�����ԣ���C��ȷ��
D���������߱�ʾ��Ӧ���������������Ĵ�С����A�Ƿ�Ӧ���Ӧ���ȣ���֮���ȣ�B��Ϊ��ܣ������������ܽ��ͣ���D����
��ѡC��
���������⿼������Ũ�ȵļ����Լ�����к͵ζ���֪ʶ�������ѶȽϴ�ע�����ͼ����������������Ե����⣮
������ͼ���漰�������ӷ���ʽ�У�Mg2++2OH-�TMg��OH��2����Al3++3OH-�TAl��OH��3����NH4++OH-�TNH3?H2O��
Al��OH��3+OH-�TAlO2-+2H2O����NaOH��Һ��Ũ��Ϊxmol/L����n��NH4+��=0.01xmol��n��Al��OH��3��=n��Al3+��=0.005xmol��
n��Mg2+��=
 =0.005xmol����n��Mg2+����n��Al3+����n��NH4+��=1��1��2��
=0.005xmol����n��Mg2+����n��Al3+����n��NH4+��=1��1��2������������þ�����ʵ�����֪��0.005xmol=0.05mol��x=10mol/L��
ͼ���м����������Ϊ20mlʱ����кͣ�PH=7���кͺ�����������ᣬPH��С����Һ�����ԣ�
ͼ�������߱�ʾ��Ӧ���������������Ĵ�С����A�Ƿ�Ӧ���Ӧ���ȣ���֮���ȣ�B��Ϊ��ܣ������������ܽ��ͣ�
���A��ͼ���漰�������ӷ���ʽ�У�Mg2++2OH-�TMg��OH��2����Al3++3OH-�TAl��OH��3����NH4++OH-�TNH3?H2O��
Al��OH��3+OH-�TAlO2-+2H2O����NaOH��Һ��Ũ��Ϊxmol/L����n��NH4+��=0.01xmol��n��Al��OH��3��=n��Al3+��=0.005xmol��
nn��Mg2+��=
 =0.005xmol����n��Mg2+����n��Al3+����n��NH4+��=1��1��2����A����
=0.005xmol����n��Mg2+����n��Al3+����n��NH4+��=1��1��2����A����B����NaOH��Һ��Ũ��Ϊxmol/L������n��Mg2+��=n��Mg��OH��2��=0.005xmol=0.05mol��x=10mol/L����B����
C��ͼ����25��ʱ����0.1mol?L-1����ζ�20mL0.1mol?L-1NaOH��Һ�������������Ϊ20mlʱ����кͣ�PH=7���кͺ�����������ᣬPH��С����Һ�����ԣ���C��ȷ��
D���������߱�ʾ��Ӧ���������������Ĵ�С����A�Ƿ�Ӧ���Ӧ���ȣ���֮���ȣ�B��Ϊ��ܣ������������ܽ��ͣ���D����
��ѡC��
���������⿼������Ũ�ȵļ����Լ�����к͵ζ���֪ʶ�������ѶȽϴ�ע�����ͼ����������������Ե����⣮

��ϰ��ϵ�д�
 Сѧ���AB��ϵ�д�
Сѧ���AB��ϵ�д� ABC����ȫ�ž�ϵ�д�
ABC����ȫ�ž�ϵ�д�
�����Ŀ
���и�������ʾ��ͼһ�µ��ǣ�������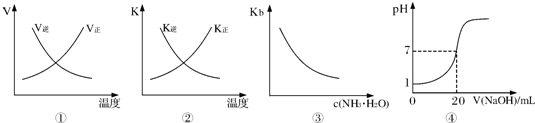

| A��ͼ�ٱ�ʾ��ӦN2��g��+O2��g��?2NO��g����H��0�����淴Ӧ���������¶ȵı仯 | B��ͼ�ڱ�ʾ��ӦN2��g��+3H2��g��?2NH3��g����H��0�����淴Ӧ��ƽ�ⳣ�����¶ȵı仯 | C��ͼ�۱�ʾ��ˮ�ĵ���ƽ�ⳣ���氱ˮŨ��c��NH3?H2O���ı仯 | D��ͼ�ܱ�ʾ25��ʱ����0.1mol?L-1NaOH��Һ�ζ�20mL 0��lmol?L-1���ᣬ��Һ��pH����������ı仯 |
����ʱ�����и�������ʾ��ͼһ�µ��ǣ�������
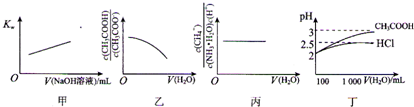

| A��ͼ�ױ�ʾ�ڴ�ˮ�м���0.01 moL?L-1 NaOH��Һ��Kw��NaOH��Һ����仯��ϵ | ||
B��ͼ�ұ�ʾ��1 mol?L-1 CH3COONa��Һ��ˮϡ�ͣ���Һ��
| ||
C��ͼ����ʾ0.1mol?L-1 NH3?H2O��ˮϡ�ͣ���Һ��
| ||
| D��ͼ����ʾ100 mL pH=2��HCl��CH3COOH��ˮϡ�͵�1000mLʱpH�仯��ˮ������ı仯��ϵ |


