��Ŀ����
2���塢�⼰�仯�����ڹ�ũҵ�������ճ�����������Ҫ��;������������ҽҩ��ʯ����ҵ���й㷺��;����ͼ��ģ�ҵ�Ʊ�����������̣�

�����������̻ش��������⣺
��1����Ӧ���з�����Ӧ�����ӷ���ʽΪSO2+Br2+2H2O=4H++2Br-+SO42-��ʹ�ñ�ˮ��Ŀ���Ǽ���Br2��HBr�ӷ�
��2�����������������������������ǹ��ˣ�
��3����Ӧ���м���Na2SO3��Ŀ���dz�ȥ��Ʒ��δ��Ӧ����壻��Ҫ��ֹ������ԭ����SO32-+2H+=SO2��+H2O�������ӷ���ʽ��ʾ��
��±�ػ�������ָ��ͬ±��ԭ��֮���Թ��ۼ�����γɵĻ����XX����±�ػ�������±�ص��ʽṹ���ơ������������������������Ӧ���ɽ���±�����������ǽ������ʷ�Ӧ������Ӧ±�������ˮ��Ӧ�ȣ��Իش��������⣺
��4���廯�⣨IBr����ˮ��Ӧ�ķ���ʽΪIBr+H2O=HBr+HIO���й���IBr�������в���ȷ����AC
A��IBr����͵�Ļ��ϼ۾�Ϊ0�� B�������෴Ӧ��IBr��ǿ������
C����IBr��ˮ�ķ�Ӧ�У����������������ǻ�ԭ�� D����NaOH��Һ��Ӧ����NaIO��NaBr��H2O
��5���ڴֵ��к���IBr��ICl������ʱ��I2��ICl��IBr�����������ڴֵ��м���һ�����ʺ��ٽ������������Ƶþ��⣬Ӧ�����������B
A��H2O B��KI C��Zn D��KCl��
���� �����̿�֪����Ӧ�Ң��з�����Ӧ SO2+Br2+2H2O=H2SO4+2HBr��������������������������Ĵ�Ʒ�����壩����Ӧ�Ң��м���Na2SO3��ԭ��Ʒ�е�Br2�����ᷴӦ����SO42- ����������������˵����ᱵ��������ɫ��Һ��������õ����µ������ᣮ
��1��Br2����ǿ�����ԣ�����Һ�н�SO2����ΪH2SO4����������ԭΪHBr��Br2����SO2�ų��ܶ�������Br2��HBr�ӷ���
��2���ɹ������̿�֪��������Ϊ���ܵ���Һ��ֵķ��룬Ӧ������������������Һ�壬Ӧ�ǹ��ˣ�
��3����Ʒ�п��ܺ���Ϊ��Ӧ��Br2��Ӧ��ȥBr2���������ƹ��˻������ᷴӦ���ɶ�������
��4��IBr��IԪ�صĻ��ϼ�Ϊ+1�ۣ�BrΪ-1�ۣ�IBr��ѧ������±�ص��ʾ��������ԣ�����ӦIBr+H2O�THBr+HIO��û��Ԫ�ػ��ϼ۵ı仯���������
��5�����õ������������Ծ��Ƶ⣬��ʹICl��IBr�е�Cl��Br��Ϊ�ѻӷ��Ե����ʣ���ʹ���е�+1�۵ĵⱻ��ԭΪI2��ͬʱ��������ʺ͵⣨I2������Ӧ��
��� �⣺��Ӧ�Ң��з�����Ӧ SO2+Br2+2H2O=H2SO4+2HBr��������������������������Ĵ�Ʒ�����壩����Ӧ�Ң��м���Na2SO3��ԭ��Ʒ�е�Br2�����ᷴӦ����SO42- ����������������˵����ᱵ��������ɫ��Һ��������õ����µ������ᣮ
��1��Br2����ǿ�����ԣ�����Һ�н�SO2����ΪH2SO4����������ԭΪHBr����Ӧ���ӷ���ʽΪSO2+Br2+2H2O=4H++2Br-+SO42-��Br2����SO2�ų��ܶ�������Br2��HBr�ӷ���ʹ�ñ�ˮ��������ϵ�¶ȣ���ֹBr2��HBr�ӷ���ʹ��Ӧ��ȫ��
�ʴ�Ϊ��SO2+Br2+2H2O=4H++2Br-+SO42-������Br2��HBr�ӷ���
��2���ɹ������̿�֪��������Ϊ���ܵ���Һ��ֵķ��룬Ӧ������������������Һ�壬Ӧ�ǹ��ˣ��õ��IJ���������©�������������ձ���
�ʴ�Ϊ�������ˣ�
��3����Ʒ�п��ܺ���Ϊ��Ӧ��Br2������Na2SO3����ȥ��Ʒ��δ��Ӧ����壬��Ҫ��ֹ�������ƹ������������ƹ����������ᷴӦ���ɶ�������Ӧ�����ӷ���ʽΪ��SO32-+2H+=SO2��+H2O��
�ʴ�Ϊ����ȥ��Ʒ��δ��Ӧ����壻SO32-+2H+=SO2��+H2O��
��4��A�������������������ڵ⣬������-1�ۡ�����+1�ۣ���A����
B���廯��Ļ�ѧ������±�ص������ƣ���IBr��IԪ�صĻ��ϼ�Ϊ+1�ۣ�IԪ�صĻ��ϼ��ڷ�Ӧ���ܽ��ͣ����ںܶѧ��Ӧ��IBr��ǿ����������B��ȷ��
C���廯����ˮ����ʱ���÷�Ӧ��û�е���ת�ƣ�������������ԭ��Ӧ����C����
D��IBr+H2O�THBr+HIO�����ɵ����ܺ�NaOH��Ӧ�������廯���NaOH��Ӧ����NaIO��NaBr��H2O����D��ȷ��
�ʴ�Ϊ��AC��
��5��A������ˮ�����µ�������������ˮ���ܽⲿ�ֵ⣬�����Ϸ��롢�ᴿ��Ŀ�ģ���A����
B������KI��������ӦΪIBr+KI=I2+KBr��ICl+KI=I2+KCl������ʱI2������KCl��KBr���룬��B��ȷ��
C������Zn���ڼ�������������ZnI��Ӱ�챻�ᴿ�����ʣ������������ԭ��C����
D������KCl�����ʲ���Ӧ���������ӵ�Ŀ�ģ���D����
�ʴ�Ϊ��B��
���� ���⿼�����ʵķ��롢�ᴿ����������ԭ��Ӧ��֪ʶ���������ʵ�����Ӧ�ã���Ŀ�Ѷ��еȣ�ע��������֪ʶ�Ļ��ۣ�

 Сѧ���AB��ϵ�д�
Сѧ���AB��ϵ�д�| A�� | 10 mL 0.1 mol•L-1��FeCl3��Һ | B�� | 20 mL 0.1 mol•L-1��MgCl2��Һ | ||
| C�� | 30 mL 0.2 mol•L-1��KCl��Һ | D�� | 100 mL 0.45 mol•L-1��KClO3��Һ |
| A�� | �����Ƿ��ж����ЧӦ������ɢϵ��Ϊ��Һ���������Һ | |
| B�� | SO2��SiO2�������������������ˮ��Ӧ������ | |
| C�� | Ư�ۡ�ˮ��������ˮ��Ϊ����� | |
| D�� | Na2O��Na2O2���Ԫ����ͬ����CO2��Ӧ����Ҳ��ȫ��ͬ |
| A�� | 0.5L��Һ������SO42-��Fe3+����ΪNA | |
| B�� | 0.5L��Һ�к���0.6NA��SO42-���� | |
| C�� | 1L��Һ�к�0.2NA��Fe3+���� | |
| D�� | 0.5L��Һ��SO42-Ũ����0.6mol/L |
| A�� | �з�ʹ�ø�Ч����������߷�Ӧ��ԭ�ϵ�ת���� | |
| B�� | ��ɫ��ѧ�ĺ������ڻ�ѧ�ϳ��н�ԭ�ӳ�����ã���Դͷ�ϼ��ٻ�������Ⱦ | |
| C�� | �ߴ��輰����������̫���ܵ�ؼ���Ϣ���ٴ���������ҪӦ�� | |
| D�� | SO2�Ǵ�����Ⱦ������Ѿ��ж�����һ������SO2��SO2�ȿ�ɱ���ֿ��������� |
| A�� | ͬŨ�ȡ�ͬ�����ǿ����ǿ����Һ��Ϻ���Һ��pH=7 | |
| B�� | ��10mL pH=a��������100mL pH=b��Ba��OH��2��Һ��Ϻ�ǡ���кͣ���a+b=13 | |
| C�� | ��pH=10��Ba��OH��2��Һ��pH=13��NaOH��Һ�������ϣ���Ϻ���Һ��pH=10.7����֪lg2=0.3�� | |
| D�� | pH=2��������pH=12�İ�ˮ�������Ϻ�������ҺpH=7 |
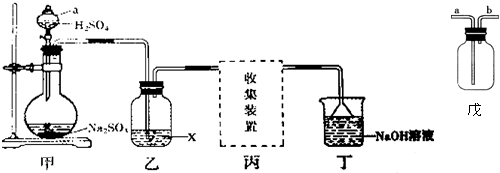

 ������I�ķ�Ӧ����ȡ����Ӧ��
������I�ķ�Ӧ����ȡ����Ӧ�� ��
��