��Ŀ����
�ס��ҡ�������Ϊ��ѧ�������ʣ����мס���Ϊ���ʣ���Ϊ���������֮�������ͼ��ʾ��ת����ϵ�� ����˵����ȷ����
����˵����ȷ����
 ����˵����ȷ����
����˵����ȷ����| A�����ס��Ҿ�Ϊ�������ʣ����������������һ���Ǽ��������� |
| B������Ϊ�������ʣ���Ϊ�ǽ������ʣ����ֻ����Mg |
| C�����ס���Ԫ����ͬ����Ԫ�أ�����Ԫ�����ڱ��Ʋ⣬��ʱ�ҵ��ʿ�����Si |
| D�����ס���Ԫ����ͬ����Ԫ�أ���÷�Ӧ�Ļ�ѧ����ʽһ��Ϊ:2F2 + 2H2O��4HF + O2 |
C
���������A. ������Al��Fe���ʣ�����Fe2O3,����Al2O3����������������������������B ����Ϊ����Fe������H2O����Ϊ�ǽ�������H2������Fe3O4������C.2C+SiO2
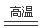 2CO��+Si����ȷ��D���ס���Ԫ����ͬ����Ԫ�أ���÷�Ӧ�Ļ�ѧ����ʽҲ����Ϊ:Zn +CuO
2CO��+Si����ȷ��D���ס���Ԫ����ͬ����Ԫ�أ���÷�Ӧ�Ļ�ѧ����ʽҲ����Ϊ:Zn +CuO ZnO + Cu.������
ZnO + Cu.������
��ϰ��ϵ�д�
 ѧϰʵ����ϵ�д�
ѧϰʵ����ϵ�д�
�����Ŀ






 R2Cu���л��ࣩ+ 2H����ˮ�ࣩ
R2Cu���л��ࣩ+ 2H����ˮ�ࣩ