��Ŀ����
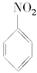
��1��N2O5�뱽����������Ӧ���ɵ��������Ľṹ��ʽ��________________
��2��2N2O5��g����4NO2��g��+O2��g��;��H��0
�ٷ�Ӧ��ƽ�������ͨ��һ������������N2O5��ת���ʽ�_______�����������С���������䡱��
���±�Ϊ��Ӧ��T1�¶��µIJ���ʵ�����ݣ�
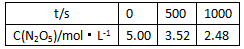
����T3�¶��£���Ӧ1000 sʱ���NO2��Ũ��Ϊ4.98 mol��L-1����T2______T1
_______________
��2���ٲ��䣻��0.00296 mol��L-1��s-1���ۡ�������С�ڡ�
��3��������N2O4+2HNO3-2e-=2N2O5+2H+

 ͬ��������ϰϵ�д�
ͬ��������ϰϵ�д� �ο�ͨ�γ̱�˼ά����������ѵ��ϵ�д�
�ο�ͨ�γ̱�˼ά����������ѵ��ϵ�д���1��N2O5�뱽����������Ӧ���ɵ��������Ľṹ��ʽ��____________________________��
��2��һ���¶��£��ں����ܱ�������N2O5�ɷ������з�Ӧ��
2N2O5��g��![]() 4NO2��g��+O2��g������H��0
4NO2��g��+O2��g������H��0
�ٷ�Ӧ��ƽ�������ͨ��һ������������N2O5��ת���ʽ�___________�������������С�����䡱��
���±�Ϊ��Ӧ��T1�¶��µIJ���ʵ�����ݣ�
t/s | 0 | 500 | 1 000 |
c��N2O5��/mol��L-1 | 5.00 | 3.52 | 2.48 |
��500 s��N2O5�ķֽ�����Ϊ__________��
����T2�¶��£���Ӧ1 000 sʱ���NO2��Ũ��Ϊ4.98 mol��L-1����T2___________T1��
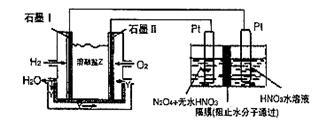
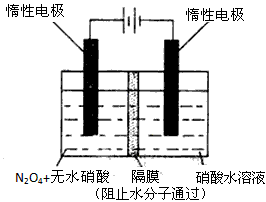
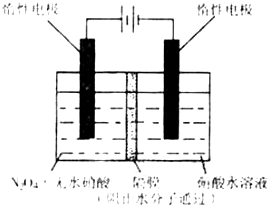
��3����ͼ��ʾװ�ÿ������Ʊ�N2O5����N2O5�ڵ��ص�_______�����ɣ���缫��ӦʽΪ____________________________________________________________________��
N2O5��һ�������������������ʺ��Ʊ��ܵ����ǵĹ�ע��![]()
![]()
![]()
![]()
![]()
![]()
![]()
��1��N2O5�뱽����������Ӧ���ɵ��������Ľṹ��ʽ�� ��
��2��һ���¶��£��ں����ܱ�������N2 O5�ɷ������з�Ӧ��
![]() 2N2O5��g�� 4NO2��g��+O2(g);��H��0
2N2O5��g�� 4NO2��g��+O2(g);��H��0
�ٷ�Ӧ��ƽ�������ͨ��һ������������N2O5��ת���ʽ� ���������С���������䡱��
���±�Ϊ��Ӧ��T1�¶��µIJ���ʵ�����ݣ�
| t/s | 0 | 500 | 1000 |
| c(N2O5)/mol��L-1 | 5.00 | 3.52 | 2.48 |
��500s��N2O5�ķֽ�����Ϊ ��
����T3�¶��£���Ӧ1 000 sʱ���NO2��Ũ��Ϊ4.98 mol��L-1����T2 T1��
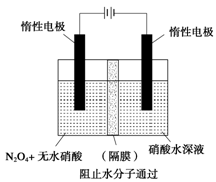 |
��3������ͼ��ʾװ�ÿ������Ʊ�N2O5����N2O5�ڵ��ص�
�����ɣ���缫��ӦʽΪ .
 ��2008?���죩N2O5��һ�������������������ʺ��Ʊ��ܵ����ǵĹ�ע��
��2008?���죩N2O5��һ�������������������ʺ��Ʊ��ܵ����ǵĹ�ע��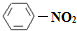
 ��X��Y��Z�Ƕ�����Ԫ�ص����ֳ��������X��ˮ��Ӧ������һ�־��л�ԭ�ԵIJ��ȶ��Ķ�Ԫ�ᣬ����Ļ�ѧʽ��
��X��Y��Z�Ƕ�����Ԫ�ص����ֳ��������X��ˮ��Ӧ������һ�־��л�ԭ�ԵIJ��ȶ��Ķ�Ԫ�ᣬ����Ļ�ѧʽ��