��Ŀ����
��һ������CO2����ͨ��500mLijNaOH��Һ�У���ַ�Ӧ����Һ�ڵ��½��������õ������ᾧˮ�İ�ɫ����A��ȡ����������ͬ��A��Ʒ�ֱ���50mL��ͬŨ�ȵ����ᷴӦ���õ�������������״���������A��������ϵְ�±���ʾ��
| �顡 �� | �� | �� | �� |
| ���������mL�� | 50 | 50 | 50 |
| A������ ��g�� | 3.80 | 5.70 | 7.60 |
| ���������mL�� | 896 | 1344 | 1344 |
��2�����ƶϣ�A�ijɷ�Ϊ______
��3����������ʵ�����Ũ��Ϊ______mol?L-1��
�⣺��1���١�����������������ʵ�����ͬ�����ڢ������ɵ�������������鶼�٣����Ե�1�����������
�ʴ�Ϊ���٣�
��2���ڢ����У�n��CO2��=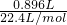 =0.04mol��
=0.04mol��
��ȫ��ΪNa2CO3��m��Na2CO3��=106g/mol��0.04mol=4.2g��
��ȫ��ΪNaHCO3��m��NaHCO3��=84g/mol��0.04mol=3.36g��
��ӦΪNa2CO3��NaHCO3�Ļ���
�ʴ�Ϊ��Na2CO3��NaHCO3��
��3���ɵڢ����֪�������A��ȫ��Ӧ���������к���xmolNa2CO3��ymolNaHCO3��
��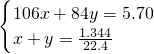
��֮�ã�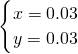
����Ԫ���غ��֪��n��HCl��=n��Na+��=2��n��Na2CO3��+n��NaHCO3��=2��0.03mol+0.03mol=0.09mol��
c��HCl��=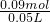 =1.8mol/L��
=1.8mol/L��
�ʴ�Ϊ��1.8��
��������1�����١�����������������ʵ�����ͬ�����ڢ������ɵ�������������鶼�٣��Դ��жϷ�Ӧ�Ĺ������⣻
��2�����ݵ�1�����ݣ������۵ķ����ж����ʵijɷ֣�
��3���ڡ������˵��������ȫ��Ӧ�����ݵ�2����Ԫ�������غ���㣮
���������⿼������ļ��㣬��Ŀ�Ѷ��еȣ�����ע��Ա������ݵķ���������ѧ�������ݷ���������������ע�����۷���Ӧ�ã�
�ʴ�Ϊ���٣�
��2���ڢ����У�n��CO2��=
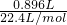 =0.04mol��
=0.04mol����ȫ��ΪNa2CO3��m��Na2CO3��=106g/mol��0.04mol=4.2g��
��ȫ��ΪNaHCO3��m��NaHCO3��=84g/mol��0.04mol=3.36g��
��ӦΪNa2CO3��NaHCO3�Ļ���
�ʴ�Ϊ��Na2CO3��NaHCO3��
��3���ɵڢ����֪�������A��ȫ��Ӧ���������к���xmolNa2CO3��ymolNaHCO3��
��
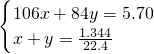
��֮�ã�
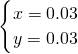
����Ԫ���غ��֪��n��HCl��=n��Na+��=2��n��Na2CO3��+n��NaHCO3��=2��0.03mol+0.03mol=0.09mol��
c��HCl��=
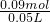 =1.8mol/L��
=1.8mol/L���ʴ�Ϊ��1.8��
��������1�����١�����������������ʵ�����ͬ�����ڢ������ɵ�������������鶼�٣��Դ��жϷ�Ӧ�Ĺ������⣻
��2�����ݵ�1�����ݣ������۵ķ����ж����ʵijɷ֣�
��3���ڡ������˵��������ȫ��Ӧ�����ݵ�2����Ԫ�������غ���㣮
���������⿼������ļ��㣬��Ŀ�Ѷ��еȣ�����ע��Ա������ݵķ���������ѧ�������ݷ���������������ע�����۷���Ӧ�ã�

��ϰ��ϵ�д�
 ��������һ���þ�ϵ�д�
��������һ���þ�ϵ�д� Сѧ��10����Ӧ����ϵ�д�
Сѧ��10����Ӧ����ϵ�д�
�����Ŀ
��һ������CO2����ͨ��500mLijNaOH��Һ�У���ַ�Ӧ����Һ�ڵ��½����������õ������ᾧˮ�İ�ɫ����A��ȡ����������ͬ��A��Ʒ�ֱ���50mL��ͬŨ�ȵ����ᷴӦ���õ�������������״���������A��������ϵ���±���ʾ��
|
�� �� |
1 |
2 |
3 |
|
���������mL�� |
50 |
50 |
50 |
|
A��������g�� |
3.80 |
5.70 |
7.60 |
|
���������mL�� |
896 |
1344 |
1344 |
��1���ϱ��е� �����ݼ����50mL����ķ�Ӧ����ʣ�࣬
��2�����ƶϣ�A�ijɷ�Ϊ
��3����������ʵ�����Ũ��Ϊ mol��L-1
 ��һ������CO2����ͨ��2L��NaOH��Һ�У���������Һ����μ���ϡ������������������Һ���ȣ�������������HCl���ʵ�����ϵ��ͼ��ʾ������������ܽ��HCl�Ļӷ�����
��һ������CO2����ͨ��2L��NaOH��Һ�У���������Һ����μ���ϡ������������������Һ���ȣ�������������HCl���ʵ�����ϵ��ͼ��ʾ������������ܽ��HCl�Ļӷ�����