��Ŀ����
8����1��pH=2��CH3COOH��Һ��ˮϡ��100����pH��4���������=��������ԭ���Ǽ�ˮϡ��ʹƽ��CH3COOH?CH3COO-+H+���ƣ������ӷ���ʽ�ͱ�Ҫ������˵������pH��ȵ�NaOH��Һ��CH3COONa��Һ���ֱ���ȵ���ͬ���¶Ⱥ�CH3COONa��Һ��pH��NaOH��Һ��pH���������=������������2��pH���ʱ����NH4Cl���ڣ�NH4��2SO4����NH4HSO4������Һ��c��NH4+���ɴ�С��˳��Ϊ��=�ڣ��ۣ�
��3����������Ϊ��ˮ������ԭ����Al3++3H2O?Al��OH��3+3H+���ɵ�����������������ˮ�����ʶ���ˮ�������ӷ���ʽ�ͱ�Ҫ������˵������
��4�������ʵ���Ũ����ͬ�������백ˮ��Ϻ���Һ�е�c��NH4+��=c��Cl-�������Ϻ���Һ�����ԣ�������������ˮ��������������=����������
��5���������NH4HSO4��Һ��NaOH��Һ��ϳ����ԣ���ԭC��NH4HSO4����ԭC��NaOH�� ���������=������������Ϻ���Һ��C��SO42-����C��NH4+�� ���������=����������
���� ��1������Ϊ���ᣬϡ�ͺ����ĵ���ƽ�������ƶ�����Һ�������ӵ����ʵ�����������������Ĵ���0.0001mol/L����������Ϊǿ����Һ��������Ϊǿ�������Σ����Ⱥ�����������Һ��pH�������䣬�������Ƶ�ˮ��̶�������ҺpH����
��2���������Ƕȿ��ǣ�һ����ͬpH�ģ�NH4��2SO4��NH4Cl��Һ��ˮ��̶��Ƿ���ȣ�����NH4HSO4����ʱ����H+ʹ��Һ�����ԣ�NH4+��ˮ�ⱻ���ƣ�
��3����������Һ�е���������ӣ�������ˮ�����������������壬��������������нϴ���������������ܹ���ˮ��
��4�������ʵ���Ũ����ͬ�������백ˮ��Ϻ���Һ�е�c��NH4+��=c��Cl-�������ݵ���غ��ж���Һ����ԣ��Ȼ����Һ�����ԣ�Ҫʹ�����Һ�����ԣ���ˮӦ����������
��5�������ߵ�Ũ�����ʱ����Ӧ��������泥���Һ��ʾ���ԣ�����������Ũ��Ӧ���Դ�Щ������������ҺΪ���ԣ����ݻ��Һ�е������غ��ж���������Ӻ�笠�����Ũ�ȴ�С��
��� �⣺��1��pH=2��CH3COOH��Һ��ˮϡ��100�������ڴ���Ϊ������ʣ����ص���ƽ�⣺CH3COOH?CH3COO-+H+��ϡ�ͺ����ĵ���̶���������Һ��pH��4��
pH��ȵ�NaOH��Һ��CH3COONa��Һ��������Ϊǿ�������Σ����������ˮ����Һ��ʾ���ԣ����Ⱥ��������ӵ�ˮ��̶���������ҺpH�������Էֱ���ȵ���ͬ���¶Ⱥ�CH3COONa��Һ��pH��NaOH��Һ��pH��
�ʴ�Ϊ��������ˮϡ��ʹƽ��CH3COOH?CH3COO-+H+���ƣ�����
��2����ͬpH�ģ�NH4��2SO4��NH4Cl��Һ�У�����ǿ�������Σ�������Һ�ʵ����Կ��ж϶���NH4+Ũ����ȣ�����NH4HSO4����ʱ����H+ʹ��Һ�����ԣ�NH4+��ˮ�ⱻ���ƣ����NH4HSO4��NH4+��Ũ��С�ڣ�NH4��2SO4������Һ��笠�����Ũ�ȴ�СΪ����=�ڣ��ۣ�
�ʴ�Ϊ����=�ڣ��ۣ�
��3����������Һ�п��Ե���������ӣ�����������Һ�з���ˮ�⣺Al3++3H2O?Al��OH��3+3H+�����ɵ�����������������ˮ�����ʶ���ˮ��
�ʴ�Ϊ��Al3++3H2O?Al��OH��3+3H+ ���ɵ�����������������ˮ�����ʶ���ˮ��
��4�������ʵ���Ũ����ͬ�������백ˮ��Ϻ���Һ�е�c��NH4+��=c��Cl-�������ݵ���غ��c��H+��=c��OH-������Һ�����ԣ��Ȼ����Һ�����ԣ�Ҫʹ�����Һ�����ԣ���ˮӦ��������������������������ˮ�������
�ʴ�Ϊ���У�����
��5���������NH4HSO4��Һ��NaOH��Һ��ϳ����ԣ������ߵ�Ũ�����ʱ����Ӧ��������泥�笠����Ӳ���ˮ�⣬��ҺΪ���ԣ���Ϊ���ԣ�����������Ũ��Ӧ���Դ�Щ������c��NH4HSO4����c��NaOH�������������غ��֪��c��SO42-��=c��NH4+��+c��NH3•H2O������c��SO42-����c��NH4+����
�ʴ�Ϊ����������
���� ���⿼��������Ũ�ȴ�С�Ƚϣ���Ŀ�Ѷ��еȣ��漰�ε�ˮ��ԭ��������ϵĶ����жϡ�������ʵĵ���ƽ�⼰��Ӱ�졢����Ũ�ȴ�С�жϵ�֪ʶ������֪ʶ��϶ࡢ�ۺ��Խ�ǿ����ֿ���ѧ���ķ������������������Ӧ��������ע�����յ���غ㡢�����غ㡢�ε�ˮ��ԭ�����ж�����Ũ�ȴ�С�е�Ӧ�÷�����

 Сѧѧϰ�ð���ϵ�д�
Сѧѧϰ�ð���ϵ�д� Сѧͬ�����������ܾ�ϵ�д�
Сѧͬ�����������ܾ�ϵ�д�| A�� | ���������� | B�� | �Ҷ���������� | ||
| C�� | C2H2��C4H6 | D�� | ����������������� |
| A�� | �������֣����ᣩ��������������̼������Һ����������ռ������ | |
| B�� | �Ҵ�����ȩ����������Cu��OH��2��Һ�����������ڣ�����ȡ��Һ | |
| C�� | �������������ᣩ���ӱ���̼������Һ���������Һ����ˮ�� | |
| D�� | �������ӣ���������������Һ���������Һ����ȥ�²�Һ�� |
| ��ѧ�� | Si-Cl | H-H | H-Cl | Si-Si |
| ����/KJ��mol | 360 | 436 | 431 | 176 |
| Ԫ�ر�� | Ԫ��������Ϣ |
| A | �䵥�����ܶ���С������ |
| B | �����Ӵ�������λ����ɣ������ǿ�������Ҫ�ɷ�֮һ |
| C | ����������B������������ͬ�ĵ��Ӳ�ṹ������B�����γ��������ӻ����� |
| D | ����������������ﶼ�����ԣ���Cͬ���� |
| E | ��Cͬ���ڣ�ԭ�Ӱ뾶�ڸ�������С |
��1��д������Ԫ�ص����ƣ�C�ƣ�E��
��2��B��C��D�ļ����Ӱ뾶�ɴ�С��˳���ǣ��û�ѧʽ��ʾ��O2-��Mg2+��Al3+
��3����д��E������A��B��C�γɵĻ����ﷴӦ�Ļ�ѧ����ʽCl2+2OH-=Cl-+ClO-+H2O
��D������A��B��C�γɵĻ������ˮ��Һ��Ӧ�����ӷ���ʽ2Al+2OH-+2H2O=2AlO2-+3H2��
��4���õ���ʽ��ʾC2B���γɹ���
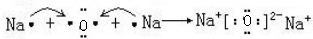 ��
�� | Ԫ�� | W | Q | M | R | X | Y |
| ԭ�Ӱ뾶/nm | 0.186 | 0.160 | 0.143 | 0.112 | 0.102 | 0.066 |
| ��Ҫ���ϼ� | +1 | +2 | +3 | +2 | +6��-2 | -2 |
| A�� | ԭ�Ӱ뾶��Y��M��Q������ԭ��������Y��M��Q | |
| B�� | M��Q��W���ǽ��������䵥�������ᷴӦ�����ʣ�M��Q��W | |
| C�� | Q2+��X2-�ĵ��Ӳ�ṹ��ͬ����X2-���Ӱ뾶����Q2+ | |
| D�� | M������������W������������ˮ���ﷴӦ |
| A�� | �����0.1mol/LHCl��0.1mol/LH2SO4�ֱ�������0.2mol/L NaOH��Һ��Ӧ | |
| B�� | ������п����п�۷ֱ�������1mol/L HCl��Ӧ | |
| C�� | �������Ũ��HCl��HNO3�ֱ����������Na2CO3��ĩ��Ӧ | |
| D�� | �����0.2mol/LHCl��0.1mol/LH2SO4������ȱ������Ʒ�ʵ�ʯ��ʯ��Ӧ |