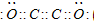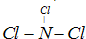��Ŀ����
����A��B��C��D��E��������������ʵ�飺
��1����ȡ0.01mol�������������ֱ�ʹ֮���ȼ�գ�����B��C��Eȼ�����õ�CO2��Ϊ448mL���������A��Dȼ�����õ�CO2����B��C��E��������
��2�����ʵ��������£�A��B��C���ܸ����������ӳɷ�Ӧ������A��ֱ��ת��ΪD��B��ת��Ϊc��E��C��ת��ΪE��
��3��B��c����ʹ��ˮ�����Ը��������Һ��ɫ����A��D��E�������ʣ�
��4������м������ʱ��A����Һ̬�巢��ȡ����Ӧ�����жϺ���д�±���
��1����ȡ0.01mol�������������ֱ�ʹ֮���ȼ�գ�����B��C��Eȼ�����õ�CO2��Ϊ448mL���������A��Dȼ�����õ�CO2����B��C��E��������
��2�����ʵ��������£�A��B��C���ܸ����������ӳɷ�Ӧ������A��ֱ��ת��ΪD��B��ת��Ϊc��E��C��ת��ΪE��
��3��B��c����ʹ��ˮ�����Ը��������Һ��ɫ����A��D��E�������ʣ�
��4������м������ʱ��A����Һ̬�巢��ȡ����Ӧ�����жϺ���д�±���
| A | B | C | D | E | |
| �ṹ��ʽ |   |
CH��CH CH��CH |
CH2=CH2 CH2=CH2 |
  |
CH3CH3 CH3CH3 |
��������ȡ0.01mol�������������ֱ�ʹ֮���ȼ�գ�����B��C��Eȼ�����õ�CO2��Ϊ448mL���������������̼�����ʵ���=
=0.02mol������B��C��E�����к���2��̼ԭ�ӣ�A��Dȼ�����õ�CO2����B��C��E����������A��D�����к���6��̼ԭ�ӣ�
���ʵ��������£�A��B��C���ܸ����������ӳɷ�Ӧ��˵�����в����ͼ���������A��ֱ��ת��ΪD��B��ת��ΪC��E��C��ת��ΪE��
B��C����ʹ��ˮ�����Ը��������Һ��ɫ����A��D��E�������ʣ�˵��B��C�к���̼̼˫����������B��C�к�������̼ԭ�ӣ�B��ת��ΪC������B����Ȳ��C����ϩ��E�����飻
����м������ʱ��A����Һ̬�巢��ȡ����Ӧ��A����6��̼ԭ�ӣ���Ϊ������A�DZ���A��ת��ΪD����D�ǻ����飮
| 0.448L |
| 22.4L/mol |
���ʵ��������£�A��B��C���ܸ����������ӳɷ�Ӧ��˵�����в����ͼ���������A��ֱ��ת��ΪD��B��ת��ΪC��E��C��ת��ΪE��
B��C����ʹ��ˮ�����Ը��������Һ��ɫ����A��D��E�������ʣ�˵��B��C�к���̼̼˫����������B��C�к�������̼ԭ�ӣ�B��ת��ΪC������B����Ȳ��C����ϩ��E�����飻
����м������ʱ��A����Һ̬�巢��ȡ����Ӧ��A����6��̼ԭ�ӣ���Ϊ������A�DZ���A��ת��ΪD����D�ǻ����飮
����⣺��ȡ0.01mol�������������ֱ�ʹ֮���ȼ�գ�����B��C��Eȼ�����õ�CO2��Ϊ448mL���������������̼�����ʵ���=
=0.02mol������B��C��E�����к���2��̼ԭ�ӣ�A��Dȼ�����õ�CO2����B��C��E����������A��D�����к���6��̼ԭ�ӣ�
���ʵ��������£�A��B��C���ܸ����������ӳɷ�Ӧ��˵�����в����ͼ���������A��ֱ��ת��ΪD��B��ת��ΪC��E��C��ת��ΪE��
B��C����ʹ��ˮ�����Ը��������Һ��ɫ����A��D��E�������ʣ�˵��B��C�к���̼̼˫����������A��D��E�в��������ͼ���B��C��E�к�������̼ԭ�ӣ�B��ת��ΪC��E��C��ת��ΪE������B����Ȳ��C����ϩ��E�����飻
����м������ʱ��A����Һ̬�巢��ȡ����Ӧ��A����6��̼ԭ�ӣ���Ϊ������A�DZ���A��ת��ΪD����E��������D�ǻ����飬
�ʴ�Ϊ�� ��CH��CH��CH2=CH2��
��CH��CH��CH2=CH2�� ��CH3CH3��
��CH3CH3��
| 0.448L |
| 22.4L/mol |
���ʵ��������£�A��B��C���ܸ����������ӳɷ�Ӧ��˵�����в����ͼ���������A��ֱ��ת��ΪD��B��ת��ΪC��E��C��ת��ΪE��
B��C����ʹ��ˮ�����Ը��������Һ��ɫ����A��D��E�������ʣ�˵��B��C�к���̼̼˫����������A��D��E�в��������ͼ���B��C��E�к�������̼ԭ�ӣ�B��ת��ΪC��E��C��ת��ΪE������B����Ȳ��C����ϩ��E�����飻
����м������ʱ��A����Һ̬�巢��ȡ����Ӧ��A����6��̼ԭ�ӣ���Ϊ������A�DZ���A��ת��ΪD����E��������D�ǻ����飬
�ʴ�Ϊ��
 ��CH��CH��CH2=CH2��
��CH��CH��CH2=CH2�� ��CH3CH3��
��CH3CH3�����������⿼�����л�����ƶϣ���ȷ�л���Ľṹ�������ǽⱾ��ؼ����ѶȲ���

��ϰ��ϵ�д�
�����Ŀ
 ����A��B��C��D��E��F���ֶ�����Ԫ�أ����ǵ�ԭ��������������D��E���⻯����ӹ��Ͷ���V�ͣ�A��B������������֮����C��������������ȣ�A�ֱܷ���B��C��D�γɵ���������ȵķ��ӣ���A��D���γɵĻ���������¾�ΪҺ̬��
����A��B��C��D��E��F���ֶ�����Ԫ�أ����ǵ�ԭ��������������D��E���⻯����ӹ��Ͷ���V�ͣ�A��B������������֮����C��������������ȣ�A�ֱܷ���B��C��D�γɵ���������ȵķ��ӣ���A��D���γɵĻ���������¾�ΪҺ̬��