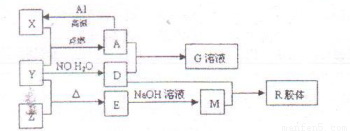
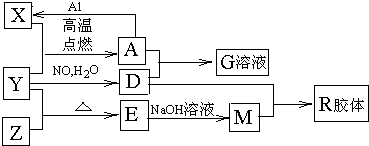
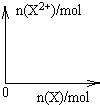��Ŀ����
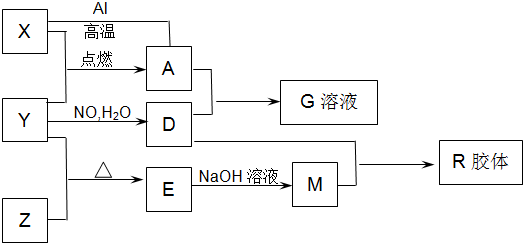
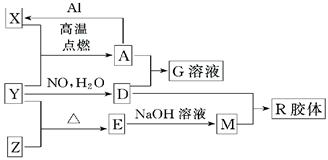
ͼ��X��Y��ZΪ���ʣ�����Ϊ��ѧ�����֮���������ת����ϵ�����ֲ�������ȥ�������У�A�׳ƴ�����������E�Dz�����ˮ�������������������ᷴӦ��

�ش��������⣺
��1��Ԫ��X��Ԫ�����ڱ��е�λ��Ϊ��________����________�塣
��2��һ�������£�Z����H2��Ӧ����ZH4 ��д��Z��NaOH��Ӧ�Ļ�ѧ����ʽ__________
��3��д����Y��NO��H2O����D��Ӧ�Ļ�ѧ����ʽ��_____________��
��4��X�ڸ���������ˮ������Ӧ����д���÷�Ӧ�Ļ�ѧ����ʽ��___________
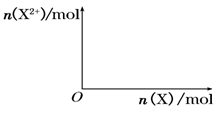
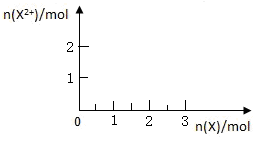
��5����4mol D��ϡ��Һ�У�����X��ĩ���������������ɵ�����ֻ��һ�֣���������ϵ�л���n(X2+)��n��X���仯��ʾ��ͼ�������n(X2+)�����ֵ��
��1��Ԫ��X��Ԫ�����ڱ��е�λ��Ϊ��________����________�塣
��2��һ�������£�Z����H2��Ӧ����ZH4 ��д��Z��NaOH��Ӧ�Ļ�ѧ����ʽ__________
��3��д����Y��NO��H2O����D��Ӧ�Ļ�ѧ����ʽ��_____________��
��4��X�ڸ���������ˮ������Ӧ����д���÷�Ӧ�Ļ�ѧ����ʽ��___________
��5����4mol D��ϡ��Һ�У�����X��ĩ���������������ɵ�����ֻ��һ�֣���������ϵ�л���n(X2+)��n��X���仯��ʾ��ͼ�������n(X2+)�����ֵ��
��1���ġ�������
��2��Si + 2NaOH + H2O ��Na2SiO3 + 2H2����
��3��4NO+3O2+2H2O=4HNO3
��4��3Fe��4H2O(g) Fe3O4��4H2
Fe3O4��4H2
��5��
��2��Si + 2NaOH + H2O ��Na2SiO3 + 2H2����
��3��4NO+3O2+2H2O=4HNO3
��4��3Fe��4H2O(g)
 Fe3O4��4H2
Fe3O4��4H2 ��5��


��ϰ��ϵ�д�
 �Ķ��쳵ϵ�д�
�Ķ��쳵ϵ�д�
�����Ŀ


 9Fe3++NO��+14H2O
9Fe3++NO��+14H2O