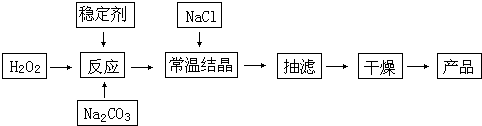
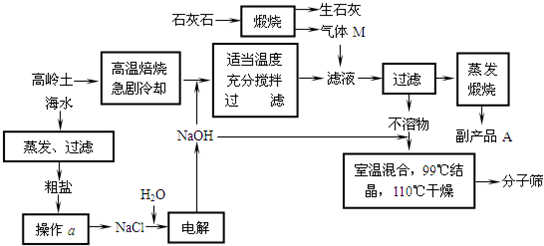
��Ŀ����
|
�����й�����Ũ�ȵıȽ���ȷ���ǵ� | |
| [����] | |
A�� |
0.1 mol��L��1��ˮ�У�c(OH��)��c(NH4+) |
B�� |
10 mL��0.02 mol��L��1HCl��Һ��10 mL��0.02 mol��L��1Ba(OH)2��Һ��ֻ�ϣ�c(Cl��)��c(Ba2+)��c(OH��)��c(H+) |
C�� |
��0.1 mol��L��1CH3COONa��0.1 mol��L��1NH4Cl�������Ϻ����Һ�У�c(H+)��c(CH3COOH)��c(NH3��H2O)��c(OH��) |
D�� |
0.1 mol��L��1ij��Ԫ�������NaHA��Һ�У�c(H+)��c(H2A)��c(A2��)��c(OH��) |
�𰸣�D

��ϰ��ϵ�д�
�����Ŀ
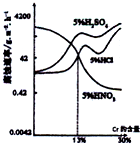 ��ͼ���������Fe-Cr�Ͻ���Cr�����仯�ĸ�ʴ��ʵ�����������й�˵����ȷ���ǣ�������
��ͼ���������Fe-Cr�Ͻ���Cr�����仯�ĸ�ʴ��ʵ�����������й�˵����ȷ���ǣ�������| A��ϡ�����Fe-Cr�Ͻ�ĸ�ʴ�Ա�ϡ�����ϡ���ᶼǿ | B������Fe���������ӣ�Fe-Cr�Ͻ���5%HNO3�е���ʴ��Խ��Խ�� | C��Cr��������13%ʱ���������Fe-Cr�Ͻ�ĸ�ʴ�����ɿ쵽����˳��ΪH2SO4��HCl=HNO3 | D��Cr����С��13%ʱ����Ϊ�������������������Ũ��������Զ�Fe-Cr�Ͻ�ĸ�ʴ����ǿ |
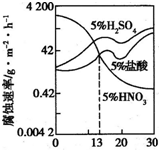 ��2010?���գ�ͼ��һϡ���Fe-Gr�Ͻ���Cr�����仯�ĸ�ʴ��ʵ�����������й�˵����ȷ���ǣ�������
��2010?���գ�ͼ��һϡ���Fe-Gr�Ͻ���Cr�����仯�ĸ�ʴ��ʵ�����������й�˵����ȷ���ǣ������� ͼ����������ijŨ��Fe2+ΪFe3+���̵�ʵ�����������й�˵����ȷ���ǣ�������
ͼ����������ijŨ��Fe2+ΪFe3+���̵�ʵ�����������й�˵����ȷ���ǣ�������