��Ŀ����
�±���Ԫ�����ڱ���һ���֣���Ա��еĢ١�=10 ����Ԫ�أ���д���пհף�
��1������ЩԪ���У���ѧ��������õ��ǣ�
��2��Ԫ�آں͢۷ǽ����Խ�������
 ��
��
��3���ܺ͢���̬�⻯����ȶ��Խ�ǿ����
��4��������������ˮ�����У�������ǿ�Ļ�����ķ���ʽ��
 ��
��
��5���õ���ʽ��ʾԪ�آٺ͢����γɻ�������γɹ��̣�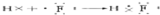
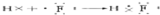 ��
��
��6���ݢޢ�����Ԫ�طֱ��γɵĵ������Ӱ뾶�ɴ�С˳��Ϊ
��7��Ԫ�آٺܰ͢�ԭ�Ӹ�����1��1�γɵĻ�����ֽ�Ļ�ѧ����ʽ
��8��д��Ԫ�آߺ͢�����������ˮ����֮�䷴Ӧ�����ӷ���ʽ
| ���� ���� |
��A | ��A | ��A | ��A | ��A | ��A | ��A | 0�� |
| һ | �� | |||||||
| �� | �� | �� | �� | �� | ||||
| �� | �� | �� | �� | �� | =10 �� | |||
Ar
Ar
����Ԫ�ط��ţ�����2��Ԫ�آں͢۷ǽ����Խ�������
C
C
����Ԫ�ط��ţ�������Ԫ�آ�ԭ�ӽṹʾ��ͼ

��3���ܺ͢���̬�⻯����ȶ��Խ�ǿ����
H20
H20
���ѧʽ������4��������������ˮ�����У�������ǿ�Ļ�����ķ���ʽ��
HClO4
HClO4
��������ǿ�Ļ�����ĵ���ʽ��

��5���õ���ʽ��ʾԪ�آٺ͢����γɻ�������γɹ��̣�
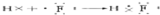
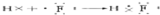
��6���ݢޢ�����Ԫ�طֱ��γɵĵ������Ӱ뾶�ɴ�С˳��Ϊ
F-��Na+��Al3+
F-��Na+��Al3+
�������ӷ��ţ�����7��Ԫ�آٺܰ͢�ԭ�Ӹ�����1��1�γɵĻ�����ֽ�Ļ�ѧ����ʽ
2H2O2
2H2O+O2��
| ||
2H2O2
2H2O+O2��
��
| ||
��8��д��Ԫ�آߺ͢�����������ˮ����֮�䷴Ӧ�����ӷ���ʽ
Al��OH��3+3H+�TAl3++3H2O
Al��OH��3+3H+�TAl3++3H2O
����������1��ϡ����������������Ϊ2��8����ѧ�����ȶ���
��2��ͬ���ڴ�����Ԫ�صķǽ���������ǿ������ԭ�Ӻ�����ӵ��Ų����������ṹʾ��ͼ��
��3��ͬ������µ�����̬�⻯����ȶ�������ǿ��
��4��ͬ���ڴ�����Ԫ�ص�����������ˮ������������ǿ������������ͬ������µ���Ԫ�ص�����������ˮ������������ǿ����������������ݻ������еĻ�ѧ������д����ʽ��
��5���������ǹ��ۼ��γɵĹ��ۻ����
��6�������������ͬ�����������Խ�࣬�뾶ԽС��
��7��˫��ˮ�ڼ��Ȼ��ö�������������ʱ���Էֽ⣻
��8�����������������ԣ����Ժ�ǿ�ᷴӦ�����κ�ˮ��
��2��ͬ���ڴ�����Ԫ�صķǽ���������ǿ������ԭ�Ӻ�����ӵ��Ų����������ṹʾ��ͼ��
��3��ͬ������µ�����̬�⻯����ȶ�������ǿ��
��4��ͬ���ڴ�����Ԫ�ص�����������ˮ������������ǿ������������ͬ������µ���Ԫ�ص�����������ˮ������������ǿ����������������ݻ������еĻ�ѧ������д����ʽ��
��5���������ǹ��ۼ��γɵĹ��ۻ����
��6�������������ͬ�����������Խ�࣬�뾶ԽС��
��7��˫��ˮ�ڼ��Ȼ��ö�������������ʱ���Էֽ⣻
��8�����������������ԣ����Ժ�ǿ�ᷴӦ�����κ�ˮ��
����⣺����Ԫ�����ڱ���Ԫ�صķֲ������֪��ΪH����ΪC����ΪN����ΪO����ΪF����ΪNa����ΪAl����ΪS����ΪCl����ΪAr��
��1��ϡ����������������Ϊ2��8����ѧ�����ȶ����ʴ�Ϊ��Ar��
��2��ͬ���ڴ�����Ԫ�صķǽ���������ǿ��C��N�Ƚϣ��ǽ����Խ�������C��Nԭ�Ӻ��������=�������������ṹʾ��ͼΪ�� ���ʴ�Ϊ��C��
���ʴ�Ϊ��C�� ��
��
��3��ͬ������µ�����̬�⻯����ȶ�������ǿ��O��S�Ƚϣ���̬�⻯����ȶ��Խ�ǿ����H20���ʴ�Ϊ��H20��
��4��ͬ���ڴ�����Ԫ�ص�����������ˮ������������ǿ������������ͬ������µ���Ԫ�ص�����������ˮ������������ǿ����������������������ǿ���Ǹ�����HClO4��������ǿ�����������ƣ��������������������Ӻ������Ӽ�ͨ����i֡���γɵ����ӻ��������ʽΪ�� ���ʴ�Ϊ��HClO4��
���ʴ�Ϊ��HClO4�� ��
��
��5������������ԭ�Ӻͷ�ԭ�Ӽ�ͨ�����ۼ��γɵĹ��ۻ�����γɹ��̿ɱ�ʾΪ��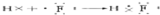 ���ʴ�Ϊ��
���ʴ�Ϊ��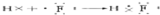 ��
��
��6��F-��Na+��Al3+������ͬ�ĺ��������������������Խ�࣬�뾶ԽС�����뾶��С˳��Ϊ��F-��Na+��Al3+��
�ʴ�Ϊ��F-��Na+��Al3+��
��7����ԭ�Ӻ���ԭ�Ӱ�ԭ�Ӹ�����1��1�γɵĻ�����˫��ˮ�ڼ��Ȼ��ö�������������ʱ���Էֽ⣬����ʽΪ��2H2O2
2H2O+O2�����ʴ�Ϊ��2H2O2
2H2O+O2����
��8����������������ˮ�����������������ȵ�����������ˮ�����Ǹ����ᣬ����ǿ�ᣬ���������������ԣ����Ժ�ǿ�ᷴӦ�����κ�ˮ��ʵ���ǣ�Al��OH��3+3H+�TAl3++3H2O���ʴ�Ϊ��Al��OH��3+3H+�TAl3++3H2O��
��1��ϡ����������������Ϊ2��8����ѧ�����ȶ����ʴ�Ϊ��Ar��
��2��ͬ���ڴ�����Ԫ�صķǽ���������ǿ��C��N�Ƚϣ��ǽ����Խ�������C��Nԭ�Ӻ��������=�������������ṹʾ��ͼΪ��
 ���ʴ�Ϊ��C��
���ʴ�Ϊ��C�� ��
����3��ͬ������µ�����̬�⻯����ȶ�������ǿ��O��S�Ƚϣ���̬�⻯����ȶ��Խ�ǿ����H20���ʴ�Ϊ��H20��
��4��ͬ���ڴ�����Ԫ�ص�����������ˮ������������ǿ������������ͬ������µ���Ԫ�ص�����������ˮ������������ǿ����������������������ǿ���Ǹ�����HClO4��������ǿ�����������ƣ��������������������Ӻ������Ӽ�ͨ����i֡���γɵ����ӻ��������ʽΪ��
 ���ʴ�Ϊ��HClO4��
���ʴ�Ϊ��HClO4�� ��
����5������������ԭ�Ӻͷ�ԭ�Ӽ�ͨ�����ۼ��γɵĹ��ۻ�����γɹ��̿ɱ�ʾΪ��
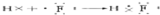 ���ʴ�Ϊ��
���ʴ�Ϊ��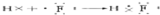 ��
����6��F-��Na+��Al3+������ͬ�ĺ��������������������Խ�࣬�뾶ԽС�����뾶��С˳��Ϊ��F-��Na+��Al3+��
�ʴ�Ϊ��F-��Na+��Al3+��
��7����ԭ�Ӻ���ԭ�Ӱ�ԭ�Ӹ�����1��1�γɵĻ�����˫��ˮ�ڼ��Ȼ��ö�������������ʱ���Էֽ⣬����ʽΪ��2H2O2
| ||
| ||
��8����������������ˮ�����������������ȵ�����������ˮ�����Ǹ����ᣬ����ǿ�ᣬ���������������ԣ����Ժ�ǿ�ᷴӦ�����κ�ˮ��ʵ���ǣ�Al��OH��3+3H+�TAl3++3H2O���ʴ�Ϊ��Al��OH��3+3H+�TAl3++3H2O��
���������⿼��ѧ��Ԫ�������ɺ�Ԫ�����ڱ����ۺ�֪ʶ��Ҫ��ѧ����ǽ̲�֪ʶ�����Ӧ�ã�

��ϰ��ϵ�д�
�����Ŀ



 NH4++OH-�����ж�NH3����ˮ���γɵ�NH3?H2O�ĺ����ṹ��
NH4++OH-�����ж�NH3����ˮ���γɵ�NH3?H2O�ĺ����ṹ�� ��4��1906���ŵ������ѧ������Ϊ�Ʊ�F2����������Ҫ���Ļ�ѧ��Ī��ɣ����Ԥ�����ȱ�������F2��Ӧ�Ʊ�ϡ�����廯�����Ԫ����
��4��1906���ŵ������ѧ������Ϊ�Ʊ�F2����������Ҫ���Ļ�ѧ��Ī��ɣ����Ԥ�����ȱ�������F2��Ӧ�Ʊ�ϡ�����廯�����Ԫ����