��Ŀ����
���Ц�H��ʾ����ȼ���ȵ���________����ʾ�����к��ȵ���________(�H1����H2����H3��)��
A��2H2(g)��O2(g)��2H2O(l)����H1
B��C(s)��1/2O2(g)��CO(g)����H2
C��CH4(g)��2O2(g)��CO2(g)��2H2O(g)����H3
D��C(s)��O2(g)��CO2(g)����H4
E��1/6C6H12O6(s)��O2(g)��CO2(g)��H2O(l)����H5
F��NaOH(s)��HCl(aq)��NaCl(aq)��H2O(l)����H6
G��2NaOH(aq)��H2SO4(aq)��Na2SO4(aq)��2H2O(l)����H7
H��CH3COOH(aq)��NaOH(aq)��CH3COONa(aq)��H2O(l)����H8
�𰸣�
������
������
|
��H4����H8 |

��ϰ��ϵ�д�
 �Ķ��쳵ϵ�д�
�Ķ��쳵ϵ�д�
�����Ŀ
(12��)(2011�������߶����)��ѧ��Ӧ�����з������ʱ仯��ͬʱ���������������ı仯�����������ı仯�������ܵ���ʽ���ֳ�����������Ӧ�ȡ����ڷ�Ӧ�������ͬ����Ӧ�ȿ��Է�Ϊ�����֣���ȼ���Ⱥ��к��ȵȡ�
(1)���Ц�H��ʾ����ȼ���ȵ���________����ʾ�����к��ȵ���________��(�H1����H2�ͦ�H3��)
| A��2H2(g)��O2(g)===2H2O(l)����H1 |
| B��C(s)��O2(g)===CO(g)����H2 |
| C��CH4(g)��2O2(g)===CO2(g)��2H2O(g)����H3 |
| D��C(s)��O2(g)===CO2(g)����H4 |
F��NaOH(aq)��HCl(aq)===NaCl(aq)��H2O(l)����H6
G��2NaOH(aq)��H2SO4(aq)===Na2SO4(aq)��2H2O(l)����H7
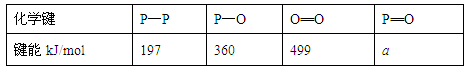
(2)��һ���о���������ѧ��Ӧ�������仯(��H)�뷴Ӧ���������ļ����йء����ܿ��Լ�����Ϊ�Ͽ�1 mol��ѧ��ʱ�������յ��������±��Dz��ֻ�ѧ���ļ������ݣ�
| ��ѧ�� | P��P | P��O | O===O | P===O |
| ����kJ/mol | 197 | 360 | 499 | x |

 ��ѧ��Ӧ���̷������ʱ仯ͬʱ�����������仯������������ʽ���ֳ������з�Ӧ�ȣ����кܶ��֣�ȼ���ȣ��к��ȵȣ�
��ѧ��Ӧ���̷������ʱ仯ͬʱ�����������仯������������ʽ���ֳ������з�Ӧ�ȣ����кܶ��֣�ȼ���ȣ��к��ȵȣ�