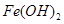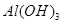��Ŀ����
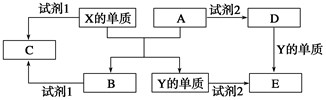
��ͼ�Ǵ�ͳ�Ĺ�ҵ�����������Ļ�������ͼ��
��ش�
��1�����������������������������Ʋ;߲������������ʱ�������ԡ����Ե�ʳ�д����������������Һ��Ӧ�����ӷ���ʽ��_____________________________________��
ʯ�����ƺ�ú�ĸ����Ʒ________(����������)����������������������ϡ�
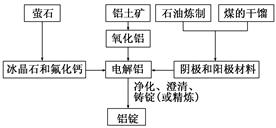
��2����ұ�������У�����������Ҫ���ڽ��и�����ԭ���Ǹü����ϲ��ϱ����ģ��������������ԭ����____________________________________(�û�ѧ����ʽ��ʾ)��
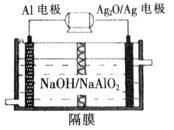
��3����ҵ��ͨ��������ڵ�MgCl2��ȡ����þ����ⷴӦ����ʽΪ________________��þ�������ǻ��ý�����Ϊʲô�ڵ��ұ�������У�һ�����Ȼ��һ�����������˵�����ɡ�________________________________________________��

��ش�
��1�����������������������������Ʋ;߲������������ʱ�������ԡ����Ե�ʳ�д����������������Һ��Ӧ�����ӷ���ʽ��_____________________________________��
ʯ�����ƺ�ú�ĸ����Ʒ________(����������)����������������������ϡ�
��2����ұ�������У�����������Ҫ���ڽ��и�����ԭ���Ǹü����ϲ��ϱ����ģ��������������ԭ����____________________________________(�û�ѧ����ʽ��ʾ)��
��3����ҵ��ͨ��������ڵ�MgCl2��ȡ����þ����ⷴӦ����ʽΪ________________��þ�������ǻ��ý�����Ϊʲô�ڵ��ұ�������У�һ�����Ȼ��һ�����������˵�����ɡ�________________________________________________��
��1��2Al��2OH����2H2O=2AlO2-��3H2����ʯī(��̼)
��2��2C��O2 2CO
2CO
��3��MgCl2(����) Mg��Cl2����MgO���ܵ�̫�ߣ���MgCl2���۵�ϵͣ��ۻ�ʱMgCl2�ܷ�����������磻AlCl3�ǹ��ۻ�����ۻ�ʱ���ܷ�������
Mg��Cl2����MgO���ܵ�̫�ߣ���MgCl2���۵�ϵͣ��ۻ�ʱMgCl2�ܷ�����������磻AlCl3�ǹ��ۻ�����ۻ�ʱ���ܷ�������
��2��2C��O2
 2CO
2CO��3��MgCl2(����)
 Mg��Cl2����MgO���ܵ�̫�ߣ���MgCl2���۵�ϵͣ��ۻ�ʱMgCl2�ܷ�����������磻AlCl3�ǹ��ۻ�����ۻ�ʱ���ܷ�������
Mg��Cl2����MgO���ܵ�̫�ߣ���MgCl2���۵�ϵͣ��ۻ�ʱMgCl2�ܷ�����������磻AlCl3�ǹ��ۻ�����ۻ�ʱ���ܷ���������1��Al��ǿ����Һ���õõ�ƫ���������������ʷ�Ӧ�����ӷ���ʽΪ2Al��2OH����2H2O=2AlO2-��3H2����
��2�������������������������ڸ�����������缫����(ʯī)��Ӧ����CO��
��2�������������������������ڸ�����������缫����(ʯī)��Ӧ����CO��

��ϰ��ϵ�д�
 ��������һ���þ�ϵ�д�
��������һ���þ�ϵ�д� Сѧ��10����Ӧ����ϵ�д�
Сѧ��10����Ӧ����ϵ�д�
�����Ŀ
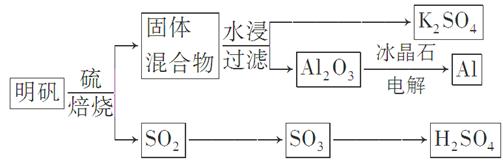 ���������Ļ�ѧ����ʽΪ��4KAl��SO4��2��12H2O��3S=2K2SO4��2Al2O3��9SO2����48H2O
���������Ļ�ѧ����ʽΪ��4KAl��SO4��2��12H2O��3S=2K2SO4��2Al2O3��9SO2����48H2O
 2SO3��g������H1����197 kJ/mol��
2SO3��g������H1����197 kJ/mol��