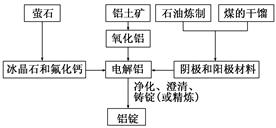
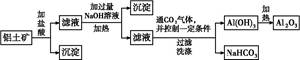
��Ŀ����
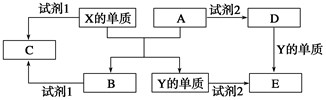
A��B��C��D��E����ѧ�γ�����5�ֻ����A��B�������Ԫ��X��Y�ĵ����������г����Ľ�����������ʼ��ת����ϵ��ͼ��ʾ��
(1)X�ĵ�����A��Ӧ�Ļ�ѧ����ʽ��_______________________________��
(2)���Լ�1��NaOH��Һ����X�ĵ������Լ�1��Ӧ�����ӷ���ʽ��
___________________________________
(3)���Լ�1���Լ�2����ϡ���ᡣ
�ټ�������D����Һ�н������ӵķ�����___________________________________��
�ڽ�����C����ˮ������Һ�����ԣ�ԭ����(�����ӷ���ʽ��ʾ)
________________________________________________��
��ij��Ч��ˮ������Y(OH)SO4�ۺϵõ��ģ���ҵ����E��ϡ�������������Ϊԭ���Ʊ�Y(OH)SO4����Ӧ����NO���ɣ��÷�Ӧ�Ļ�ѧ����ʽ��_____________________��

(1)X�ĵ�����A��Ӧ�Ļ�ѧ����ʽ��_______________________________��
(2)���Լ�1��NaOH��Һ����X�ĵ������Լ�1��Ӧ�����ӷ���ʽ��
___________________________________
(3)���Լ�1���Լ�2����ϡ���ᡣ
�ټ�������D����Һ�н������ӵķ�����___________________________________��
�ڽ�����C����ˮ������Һ�����ԣ�ԭ����(�����ӷ���ʽ��ʾ)
________________________________________________��
��ij��Ч��ˮ������Y(OH)SO4�ۺϵõ��ģ���ҵ����E��ϡ�������������Ϊԭ���Ʊ�Y(OH)SO4����Ӧ����NO���ɣ��÷�Ӧ�Ļ�ѧ����ʽ��_____________________��
(1)Fe2O3��2Al 2Fe��Al2O3
2Fe��Al2O3
(2)2Al��2OH����2H2O=2AlO2����3H2��
(3)��ȡ����D����Һ���Թ��У��μӼ���KSCN��Һ����Һ��Ѫ��ɫ��֤����Fe3��
��Al3����3H2O Al(OH)3��3H��
Al(OH)3��3H��
��2FeSO4��2NaNO2��H2SO4=2Fe(OH)SO4��Na2SO4��2NO��
 2Fe��Al2O3
2Fe��Al2O3(2)2Al��2OH����2H2O=2AlO2����3H2��
(3)��ȡ����D����Һ���Թ��У��μӼ���KSCN��Һ����Һ��Ѫ��ɫ��֤����Fe3��
��Al3����3H2O
 Al(OH)3��3H��
Al(OH)3��3H����2FeSO4��2NaNO2��H2SO4=2Fe(OH)SO4��Na2SO4��2NO��
A��B�������Ԫ��X��Y�ĵ����������г����Ľ��������ͼ��֪A��Fe2O3��X�ĵ�����Al��B��Al2O3��Y�ĵ�����Fe�����Լ�1��NaOH��Һ����CΪNaAlO2�����Լ�1���Լ�2����ϡ���ᣬ��D����Һ�к�Fe3��������KSCN��Һ���飬C����Һ�к�Al3������ˮ�⣬EΪFeSO4��

��ϰ��ϵ�д�
 ȫ�ܲ��һ���þ�ϵ�д�
ȫ�ܲ��һ���þ�ϵ�д� ��ɢ˼ά�¿���ϵ�д�
��ɢ˼ά�¿���ϵ�д�
�����Ŀ