ЬтФПФкШн
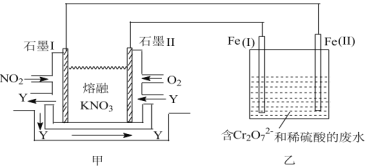
ЁОЬтФПЁПСЌЖўбЧСђЫсФЦ(Na2S2O4)ЪЧгЁШОЙЄвЕЕФвЛжжГЃгУдСЯЃЌСЌЖўбЧСђЫсФЦгжГЦБЃЯеЗлЃЌПЩШмгкЫЎЃЌЕЋВЛШмгкМзДМЃЌЦфЙЬЬхЪмШШЁЂгіЫЎЖМЛсЗЂЩњЗДгІЗХГіДѓСПЕФШШЃЌЩѕжСв§Ц№ШМЩеЃЌЙЄвЕжЦБИСїГЬШчЭМЃК
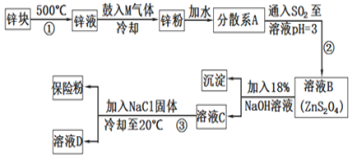
ЧыЛиД№ЯТСаЮЪЬтЃК
ЃЈ1ЃЉБЃДцСЌЖўбЧСђЫсФЦЕФзЂвтЪТЯю___(ШЮаДвЛЯю)ЁЃ
ЃЈ2ЃЉЙФШыЕФЦјЬхMЮЊПеЦјЕФвЛжжжївЊГЩЗжЃЌдђMЕФЕчзгЪНЮЊ___ЃЛ
ЃЈ3ЃЉСїГЬЂкЕФЛЏбЇЗНГЬЪНЮЊ__ЁЃ
ЃЈ4ЃЉСїГЬЂлЕФЗжРыЗНЗЈВНжшЮЊЙ§ТЫЁЂЯДЕгЁЂИЩдяЁЃЦфжаЯДЕгЫљгУЪдМСЪЧ___ЃЛЪЕбщЪБМгШыNaClЙЬЬхЕФзїгУЪЧ__ЁЃ
ЃЈ5ЃЉСЌЖўбЧСђЫсФЦПЩвдгУгкГ§ШЅЗЯЫЎжаЕФжиИѕЫсИљРызг(Cr2O72-БЛзЊЛЏЮЊCr3+ЃЌS2O42-БЛзЊЛЏЮЊSO42-)ЃЌаДГіИУЗДгІЕФРызгЗНГЬЪН___ЁЃ
ЁОД№АИЁПУмЗтЁЂвѕСЙДІБЃДц ![]() Zn+2SO2=ZnS2O4 МзДМ НЕЕЭNa2S2O4ЕФШмНтЖШЛђдіДѓNa+ХЈЖШЃЌБугкNa2S2O4НсОЇЮіГі Cr2O72-+S2O42-+6H+=2Cr3++2SO42- +3H2O
Zn+2SO2=ZnS2O4 МзДМ НЕЕЭNa2S2O4ЕФШмНтЖШЛђдіДѓNa+ХЈЖШЃЌБугкNa2S2O4НсОЇЮіГі Cr2O72-+S2O42-+6H+=2Cr3++2SO42- +3H2O
ЁОНтЮіЁП
аППщМгШШШкЛЏЃЌЯђаПвКжаЙФШыMЦјЬхЃЌЪЙвКЬЌаПЮэЛЏЃЌРфШДЕУЕНаПЗлЃЌМгЫЎаЮГЩЗжЩЂЯЕЃЌдйЭЈШыЖўбѕЛЏСђЗДгІЕУЕНZnS2O4ЃЌМгШыNaOHШмвКЗДгІЕУЕНЧтбѕЛЏаПГСЕэЁЂNa2S2O4ЃЌзюКѓМгШыNaClНЕЕЭNa2S2O4ЕФШмНтЖШЃЌЮіГіNa2S2O4ЃЌШмвКDжаКЌгаNaClЃЌОнДЫЗжЮізїД№ЁЃ
ЃЈ1ЃЉвђЮЊЬтФПжаЙигкБЃЯеЗлЫЕУїЮЊЃКЦфЙЬЬхЪмШШЁЂгіЫЎЖМЛсЗЂЩњЗДгІЗХГіДѓСПЕФШШЃЌЩѕжСв§Ц№ШМЩеЁЃЫљвдБЃДцЪБгІИУУмЗтБЃДцЃЌБмУтЪмШШЃЌЭЌЪБвВгІИУгыЦфЫћвзШМЮяЗжРыЃЛ
ЃЈ2ЃЉЙФШыЕФЦјЬхMЮЊПеЦјЕФвЛжжжївЊГЩЗжЃЌдђMжЛФмЪЧЕЊЦјЃЌЦфЕчзгЪНЮЊ![]() ЃЛ
ЃЛ
ЃЈ3ЃЉСїГЬЂкЕФФПЕФЪЧНЋЕЅжЪаПзЊЛЏЮЊZnS2O4ЃЌЫљвдЯђZnЗлжаЭЈШыSO2ЃЌЦфЛЏбЇЗНГЬЪНЮЊЃКZn+2SO2 = ZnS2O4ЃЛ
ЃЈ4ЃЉЯДЕгВњЦЗЪБЃЌПМТЧЕНЬтФПжаЖдгкБЃЯеЗлЕФУшЪіЃКПЩШмгкЫЎЃЌЕЋВЛШмгкМзДМЃЌЫљвдгУМзДМЯДЕгПЩвдЗРжЙВњЦЗЕФЫ№ЪЇЃЛЪЕбщжаМгШыТШЛЏФЦЕФФПЕФЪЧРћгУЭЌРызгаЇгІЃЌНЕЕЭNa2S2O4ЕФШмНтЖШЃЌвдБугкЦфЮіГіЃЌЙЪД№АИЮЊЃКМзДМЃЛНЕЕЭNa2S2O4ЕФШмНтЖШЛђдіДѓNa+ХЈЖШЃЌБугкNa2S2O4НсОЇЮіГіЃЛ
ЃЈ5ЃЉСЌЖўбЧСђЫсФЦПЩвдгУгкГ§ШЅЗЯЫЎжаЕФжиИѕЫсИљРызг(Cr2O72-БЛзЊЛЏЮЊCr3+ЃЌS2O42-БЛзЊЛЏЮЊSO42-)ЃЌгЩЩЯЕУЕНВПЗжЗДгІЃКCr2O72-+S2O42- Ёњ Cr3+ +SO42-ЃЌCr2O72-ЛЏКЯМлНЕЕЭ6МлЃЈ2ИіCrЃЌУПИіНЕЕЭ3МлЃЉЃЌS2O42-ЛЏКЯМлЩ§Ип6МлЃЈ2ИіSЃЌУПИіЩ§Ип3МлЃЉЃЌЫљвдСНИіРызгЕФЯЕЪ§ЖМЪЧ1ЃЌЗНГЬЪНБфЮЊCr2O72-+S2O42- Ёњ 2Cr3+ + 2SO42-ЃЌдйИљОндзгИіЪ§ЪиКуКЭЕчКЩЪиКуЕУЕНЃКCr2O72-+S2O42-+6H+ = 2Cr3+ +2SO42- +3H2OЁЃ

 УћаЃПЮЬУЯЕСаД№АИ
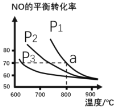
УћаЃПЮЬУЯЕСаД№АИЁОЬтФПЁПt ЁцЪБЃЌдкЬхЛ§ВЛБфЕФУмБеШнЦїжаЗЂЩњЗДгІЃКX(g)+3Y(g)![]() 2Z(g)ЃЌИїзщЗждкВЛЭЌЪБПЬЕФХЈЖШШчЯТБэЃЌЯТСаЫЕЗЈе§ШЗЕФЪЧ
2Z(g)ЃЌИїзщЗждкВЛЭЌЪБПЬЕФХЈЖШШчЯТБэЃЌЯТСаЫЕЗЈе§ШЗЕФЪЧ
ЮяжЪ | X | Y | Z |
ГѕЪМХЈЖШ/molЁЄL-1 | 0.1 | 0.2 | 0 |
2 minФЉХЈЖШ/molЁЄL-1 | 0.08 | a | b |
ЦНКтХЈЖШ/molЁЄL-1 | 0.05 | 0.05 | 0.1 |
A. ЦНКтЪБЃЌXЕФзЊЛЏТЪЮЊ20%
B. t ЁцЪБЃЌИУЗДгІЕФЦНКтГЃЪ§ЮЊ40
C. ЧА2 minФкЃЌгУYЕФБфЛЏСПБэЪОЕФЦНОљЗДгІЫйТЪv(Y)="0.03" molЁЄL-1ЁЄmin-1
D. діДѓЦНКтКѓЕФЬхЯЕбЙЧПЃЌvе§діДѓЃЌvФцМѕаЁЃЌЦНКтЯђе§ЗДгІЗНЯђвЦЖЏ
ЁОЬтФПЁПЛюадЬППЩДІРэДѓЦјЮлШОЮяNOЁЃЮЊФЃФтИУЙ§ГЬЃЌTЁцЪБЃЌдк3LУмБеШнЦїжаМгШыNOКЭЛюадЬПЗлЃЌЗДгІЬхЯЕжаИїЮяжЪЕФСПБфЛЏШчЯТБэЫљЪОЁЃЯТСаЫЕЗЈе§ШЗЕФЪЧ
ЛюадЬП/mol | NO/mol | X/mol | Y/mol | |
Ц№ЪМЪБ | 2.030 | 0.100 | 0 | 0 |
10minДяЦНКт | 2.000 | 0.040 | 0.030 | 0.030 |
A. XвЛЖЈЪЧN2ЃЌYвЛЖЈЪЧCO2
B. 10minКѓдіДѓбЙЧПЃЌNOЕФЮќЪеТЪдіДѓ
C. 10minКѓМгШыЛюадЬПЃЌЦНКтЯђе§ЗДгІЗНЯђвЦЖЏ
D. 0~10minЕФЦНОљЗДгІЫйТЪv(NO)=0.002 mol/(Lmin)
ЁОЬтФПЁПвдЙшдхЭСЮЊдиЬхЕФЮхбѕЛЏЖўЗАЃЈV2O5ЃЉЪЧНгДЅЗЈЩњГЩСђЫсЕФДпЛЏМСЁЃДгЗЯЗАДпЛЏМСжаЛиЪеV2O5МШБмУтЮлШОЛЗОГгжгаРћгкзЪдДзлКЯРћгУЁЃЗЯЗАДпЛЏМСЕФжївЊГЩЗжЮЊЃК
ЮяжЪ | V2O5 | V2O4 | K2SO4 | SiO2 | Fe2O3 | Al2O3 |
жЪСПЗжЪ§/% | 2.2~2.9 | 2.8~3.1 | 22~28 | 60~65 | 1~2 | ЃМ1 |
вдЯТЪЧвЛжжЗЯЗАДпЛЏМСЛиЪеЙЄвеТЗЯпЃК
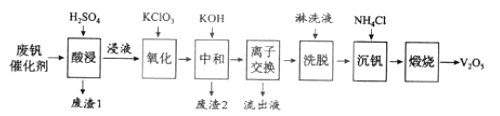
ЛиД№ЯТСаЮЪЬтЃК
ЃЈ1ЃЉЁАЫсНўЁБЪБV2O5зЊЛЏЮЊVO2+ЃЌЗДгІЕФРызгЗНГЬЪНЮЊ___________ЃЌЭЌЪБV2O4зЊГЩVO2+ЁЃЁАЗЯдќ1ЁБЕФжївЊГЩЗжЪЧ__________________ЁЃ
ЃЈ2ЃЉЁАбѕЛЏЁБжагћЪЙ3 molЕФVO2+БфЮЊVO2+ЃЌдђашвЊбѕЛЏМСKClO3жСЩйЮЊ______molЁЃ
ЃЈ3ЃЉЁАжаКЭЁБзїгУжЎвЛЪЧЪЙЗАвдV4O124аЮЪНДцдкгкШмвКжаЁЃЁАЗЯдќ2ЁБжаКЌга_______ЁЃ
ЃЈ4ЃЉЁАРызгНЛЛЛЁБКЭЁАЯДЭбЁБПЩМђЕЅБэЪОЮЊЃК4ROH+ V4O124![]() R4V4O12+4OHЃЈвдROHЮЊЧПМюадвѕРызгНЛЛЛЪїжЌЃЉЁЃЮЊСЫЬсИпЯДЭбаЇТЪЃЌСмЯДвКгІИУГЪ_____адЃЈЬюЁАЫсЁБЁАМюЁБЁАжаЁБЃЉЁЃ
R4V4O12+4OHЃЈвдROHЮЊЧПМюадвѕРызгНЛЛЛЪїжЌЃЉЁЃЮЊСЫЬсИпЯДЭбаЇТЪЃЌСмЯДвКгІИУГЪ_____адЃЈЬюЁАЫсЁБЁАМюЁБЁАжаЁБЃЉЁЃ
ЃЈ5ЃЉЁАСїГівКЁБжабєРызгзюЖрЕФЪЧ________ЁЃ
ЃЈ6ЃЉЁАГСЗАЁБЕУЕНЦЋЗАЫсяЇЃЈNH4VO3ЃЉГСЕэЃЌаДГіЁАьбЩеЁБжаЗЂЩњЗДгІЕФЛЏбЇЗНГЬЪН____________ЁЃ