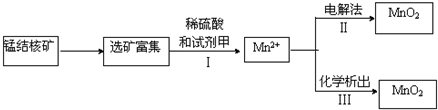
��Ŀ����
��0.2mol Mg��Al�Ļ������������ȫ�ܽ⣬�ų���״����5.6L���壬Ȼ���ٵ���2mol?L-1 NaOH��ҺV mL����ش�
��1����û������Mg�����ٷ���Ϊ�� %��С�������һλ����
��2�����û����������ǡ����ȫ��Ӧ���ڵμ�NaOH��Һ�����У���ʹMg2+��Al3+�պó�����ȫ�������NaOH��Һ�����V= mL��
��3�����û��������200mL 3mol/L�������NaOH��Һ��ʹ���ó�������Al��OH��3�������NaOH��Һ�����V����Ϊ�� mL��
��1����û������Mg�����ٷ���Ϊ��
��2�����û����������ǡ����ȫ��Ӧ���ڵμ�NaOH��Һ�����У���ʹMg2+��Al3+�պó�����ȫ�������NaOH��Һ�����V=
��3�����û��������200mL 3mol/L�������NaOH��Һ��ʹ���ó�������Al��OH��3�������NaOH��Һ�����V����Ϊ��
���㣺�йػ���ﷴӦ�ļ���,þ��������Ҫ������
ר�⣺������
��������1����Ͻ���MgΪxmol��AlΪymol�����ݶ�����������������������з��̼��㣬��������Mg������������
��2��������Ӧ��Mg2++20H-=Mg��OH��2����Al3++30H-=Al��OH��3�������ݷ���ʽ������Ҫ�������Ƶ����ʵ������ٸ���V=
���㣻
��3������Mg��Al���������200mL 3mol/L�������NaOH��Һ��ʹ���ó�������Al��OH��3��˵��AlԪ����ƫ��������ʽ���ڣ��ʴ�ʱ��Һ������ΪNaCl��NaAlO2�������������غ��֪n��NaOH��=n��NaCl��+n��NaAlO2�������������غ�n��NaCl��=n��HCl������AlԪ���غ�n��NaAlO2��=n��Al�����ݴ˼���n��NaOH�����ٸ���V=
���㣮
��2��������Ӧ��Mg2++20H-=Mg��OH��2����Al3++30H-=Al��OH��3�������ݷ���ʽ������Ҫ�������Ƶ����ʵ������ٸ���V=
| n |
| c |
��3������Mg��Al���������200mL 3mol/L�������NaOH��Һ��ʹ���ó�������Al��OH��3��˵��AlԪ����ƫ��������ʽ���ڣ��ʴ�ʱ��Һ������ΪNaCl��NaAlO2�������������غ��֪n��NaOH��=n��NaCl��+n��NaAlO2�������������غ�n��NaCl��=n��HCl������AlԪ���غ�n��NaAlO2��=n��Al�����ݴ˼���n��NaOH�����ٸ���V=
| n |
| c |
���
�⣺��1��þ���Ͻ������ᷢ����Mg+2HCl=MgCl2+H2����2Al+6HCl=2AlCl3+3H2������Ͻ���MgΪxmol��AlΪymol����
����֮��x=0.1��y=0.1��
�ʻ������Mg������=0.1mol��24g/mol=2.4g��Al������=0.1mol��27g/mol=2.7g��
�ʻ������Mg����������=
��100%=47.1%��
�ʴ�Ϊ��47.1%��
��2������Mg��Al�Ļ����������ǡ����ȫ��Ӧ���ڵμ�NaOH��Һ�����У���ʹMg2+��Al3+�պó�����ȫ��
������Mg2++20H-=Mg��OH��2����Al3++30H-=Al��OH��3����
����Ҫn��NaOH��=2n��Mg��+3n��Al��=2��0.1mol+3��0.1mol=0.5mol��
V��NaOH��=
=0.25L=250mL��
�ʴ�Ϊ��250��
��3������Mg��Al���������200mL 3mol/L�������NaOH��Һ��ʹ���ó�������Al��OH��3��˵��AlԪ����ƫ��������ʽ���ڣ��ʴ�ʱ��Һ������ΪNaCl��NaAlO2��
�����������غ��֪n��NaOH��=n��NaCl��+n��NaAlO2�������������غ�n��NaCl��=n��HCl������AlԪ���غ�n��NaAlO2��=n��Al������n��NaOH��=n��HCl��+n��Al��=0.2L��3mol/L+0.1mol=0.7mol����V��NaOH��=
=0.35L=350mL��
�ʴ�Ϊ��350��
|
�ʻ������Mg������=0.1mol��24g/mol=2.4g��Al������=0.1mol��27g/mol=2.7g��
�ʻ������Mg����������=
| 2.4g |
| 2.4g+2.7g |
�ʴ�Ϊ��47.1%��
��2������Mg��Al�Ļ����������ǡ����ȫ��Ӧ���ڵμ�NaOH��Һ�����У���ʹMg2+��Al3+�պó�����ȫ��
������Mg2++20H-=Mg��OH��2����Al3++30H-=Al��OH��3����
����Ҫn��NaOH��=2n��Mg��+3n��Al��=2��0.1mol+3��0.1mol=0.5mol��
V��NaOH��=
| 0.5mol |
| 2mol/L |
�ʴ�Ϊ��250��
��3������Mg��Al���������200mL 3mol/L�������NaOH��Һ��ʹ���ó�������Al��OH��3��˵��AlԪ����ƫ��������ʽ���ڣ��ʴ�ʱ��Һ������ΪNaCl��NaAlO2��
�����������غ��֪n��NaOH��=n��NaCl��+n��NaAlO2�������������غ�n��NaCl��=n��HCl������AlԪ���غ�n��NaAlO2��=n��Al������n��NaOH��=n��HCl��+n��Al��=0.2L��3mol/L+0.1mol=0.7mol����V��NaOH��=
| 0.7mol |
| 2mol/L |
�ʴ�Ϊ��350��
���������⿼�黯ѧ����ʽ�ļ��㡢�������㣬������ѧ���ķ��������ͼ��������Ŀ��飬��3��ע�����Ԫ���غ���㣬�Ѷ��еȣ�

��ϰ��ϵ�д�
�����Ŀ
��֪��
��2H2��g��+O2��g����2H2O��g��+483.6kJ��
��H2��g��+S��g����H2S��g��+20.1kJ��
�����ж�һ����ȷ���ǣ�������
��2H2��g��+O2��g����2H2O��g��+483.6kJ��
��H2��g��+S��g����H2S��g��+20.1kJ��
�����ж�һ����ȷ���ǣ�������
| A��1mol������ȫȼ������246.8 kJ |
| B��1molˮ������1mol������������221.7 kJ |
| C���ɢ٢�֪��ˮ�����ȶ���С������ |
| D������Ӧ���и��ù�̬���Ƚ�С��20.1 kJ |
���и������У����������ö��Բ��ϵ��ǣ�������
| A���ȼҵ |
| B����������������ƽ����� |
| C����� |
| D�������������Һ�������������� |