��Ŀ����
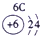
�±�ΪԪ�����ڱ���һ���֣������Ԫ�آ٣����ڱ��е�λ�ã�����ѧ�����ش��������⣺
| �� ���� | IA |
| 0 | |||||
| 1 | �� | ��A | ��A | ��A | ��A | ��A | ��A |
|
| 2 |
|
|
| �� | �� | �� |
|
|
| 3 | �� |
| �� | �� |
|
| �� |
|
��1���ܡ��ݡ���ԭ�Ӱ뾶�ɴ�С��˳��Ϊ����Ԫ�ط��š�_______________________��
��2���ڡ��ۡ��ߵ���ۺ������������ǿ������˳����[�ѧʽ]________________��
��3���١��ܡ��ݡ����е�ijЩԪ�ؿ��γɼȺ����Ӽ��ֺ����Թ��ۼ��Ļ����д������һ�ֻ�����ĵ���ʽ��____________________��
(4) �ɢں͢���ɣ��Ңںܵ͢�������Ϊ3��8�Ļ�����Ľṹʽ�� ��
���к�________����������ԡ��Ǽ��ԡ���ͬ������________����
��������ݵ�ͬ������������Ԫ�صĵ��ʷ�Ӧ�Ļ�ѧ����ʽΪ __
(5) ������ݵ�����������ˮ���ﷴӦ�����ӷ���ʽΪ ��
��1��Na>Al>O����2��HNO3>H2CO3>H2SiO3(H4SiO4)��
![]() ��3��
��3��
��4��O=C=O,���ԣ��Ǽ��� 2Mg + CO2![]() 2MgO +C
2MgO +C
��5��2Al +2OH- + 2H2O 2AlO2- + 3H2����

 ����ѧ����ϵ�д�
����ѧ����ϵ�д�







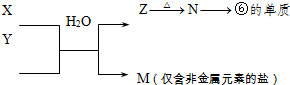
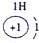
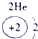
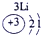
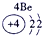
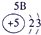
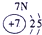
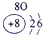
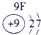
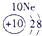
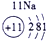





 ��ʾ����
��ʾ����