��Ŀ����
�±�ΪԪ�����ڱ���һ���֣������Ԫ�آ�-���ڱ��е�λ�ã��û�ѧ����ش��������⣺
��1���ܡ��ݡ���ԭ�Ӱ뾶�ɴ�С��˳��Ϊ______����Ԫ�ط��ű�ʾ����
��2���ڡ��ۡ��ߵ���ۺ������������ǿ������˳����______��
��3���١��ܡ��ݡ����е�ijЩԪ�ؿ��γɼȺ����Ӽ��ֺ����ۼ��Ļ����д������һ�ֻ�����ĵ���ʽ��______��
��4���ɱ�������Ԫ�ص�ԭ�Ӱ�1��1��ɵij���Һ̬�������ϡҺ�ױ����ֽ⣬��ʹ�õĴ���Ϊ������ţ�______��
a��MnO2 b��AlCl3 c��Na2SO3 d��KMnO4
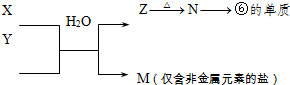
��5���ɱ���Ԫ���γɵij�������X��Y��Z��M��N�ɷ������·�Ӧ��

X��Һ��Y��Һ��Ӧ�����ӷ���ʽΪ______��N���ĵ��ʵĻ�ѧ����ʽΪ______��
��6����֪�ƻ�1mol N��N����H-H����N-H���ֱ���Ҫ���յ�����Ϊ946kJ��436kJ��391kJ����1molN2��ȫ��Ӧת��ΪNH3��______������ա��ų���������______ kJ��
| �� ���� |
IA | 0 | ||||||
| 1 | �� | ��A | ��A | ��A | ��A | ��A | ��A | |
| 2 | �� | �� | �� | |||||
| 3 | �� | �� | �� | �� | ||||
��2���ڡ��ۡ��ߵ���ۺ������������ǿ������˳����______��
��3���١��ܡ��ݡ����е�ijЩԪ�ؿ��γɼȺ����Ӽ��ֺ����ۼ��Ļ����д������һ�ֻ�����ĵ���ʽ��______��
��4���ɱ�������Ԫ�ص�ԭ�Ӱ�1��1��ɵij���Һ̬�������ϡҺ�ױ����ֽ⣬��ʹ�õĴ���Ϊ������ţ�______��
a��MnO2 b��AlCl3 c��Na2SO3 d��KMnO4
��5���ɱ���Ԫ���γɵij�������X��Y��Z��M��N�ɷ������·�Ӧ��

X��Һ��Y��Һ��Ӧ�����ӷ���ʽΪ______��N���ĵ��ʵĻ�ѧ����ʽΪ______��
��6����֪�ƻ�1mol N��N����H-H����N-H���ֱ���Ҫ���յ�����Ϊ946kJ��436kJ��391kJ����1molN2��ȫ��Ӧת��ΪNH3��______������ա��ų���������______ kJ��
����Ԫ�������ڱ��е�λ�ÿ��ж�Ԫ�ص����࣬��ΪHԪ�أ���ΪCԪ�أ���ΪNԪ�أ���ΪOԪ�أ���ΪNaԪ�أ���ΪAlԪ�أ���ΪSiԪ�أ���ΪClԪ�أ�
��1�����Ӳ�Խ��뾶Խ������O�뾶��С��ͬ����Ԫ��ԭ�Ӱ뾶��������С����Na��Al���ʴ�Ϊ��Na��Al��O��
��2��Ԫ�����ڱ��У�ͬ���ڴ�����Ԫ�صķǽ���������ǿ��ͬ������ϵ���Ԫ�صķǽ�������������ǽ����Ե�ǿ��˳��ΪN��C��Si���ǽ�����Խǿ����Ӧ����������ˮ���������Խǿ����������HNO3��H2CO3��H2SiO3���ʴ�Ϊ��HNO3��H2CO3��H2SiO3��
��3���١��ܡ��ݡ����е�ijЩԪ�ؿ��γɼȺ����Ӽ��ֺ����Թ��ۼ��Ļ�������NaOH��NaClO��NaClO3�����ʣ��������ӻ�����ʴ�Ϊ��

��
��4������������MnO2��FeCl3���������������������ֽ�������������Na2SO3���л�ԭ�ԣ���������ⷴӦ���������ᣬ������ؾ��������ԣ����������������������������������������Ȼ�����ˮ�ķֽ���Ӱ�죬�������������ʴ�Ϊ��a��
��5����

��Ӧת����֪����ΪAlԪ�أ���NΪAl2O3��ZΪAl��OH��3��MΪ�����ǽ���Ԫ�ص��Σ�ӦΪ��Σ���ӦX+YӦ�ǰ�����������ˮ��Һ�еķ�Ӧ��X��Һ��Y��Һ��Ӧ�����ӷ���ʽΪAl3++3NH3?H2O=Al��OH��3��+3NH4+����ҵұ�����õ���������ķ�������Ӧ����ʽΪ2Al2O3�����ڣ�
4Al+3O2����
�ʴ�Ϊ��Al3++3NH3?H2O=Al��OH��3��+3NH4+��2Al2O3�����ڣ�
4Al+3O2����
��6����Ӧ��=���յ�������-�ų���������������1mol N2����NH3�ķ�Ӧ��=946kJ+3��436 kJ-2��3��391kJ=-92kJ�������Ƿų��������ʴ�Ϊ���ų���92��
��1�����Ӳ�Խ��뾶Խ������O�뾶��С��ͬ����Ԫ��ԭ�Ӱ뾶��������С����Na��Al���ʴ�Ϊ��Na��Al��O��
��2��Ԫ�����ڱ��У�ͬ���ڴ�����Ԫ�صķǽ���������ǿ��ͬ������ϵ���Ԫ�صķǽ�������������ǽ����Ե�ǿ��˳��ΪN��C��Si���ǽ�����Խǿ����Ӧ����������ˮ���������Խǿ����������HNO3��H2CO3��H2SiO3���ʴ�Ϊ��HNO3��H2CO3��H2SiO3��
��3���١��ܡ��ݡ����е�ijЩԪ�ؿ��γɼȺ����Ӽ��ֺ����Թ��ۼ��Ļ�������NaOH��NaClO��NaClO3�����ʣ��������ӻ�����ʴ�Ϊ��

��
��4������������MnO2��FeCl3���������������������ֽ�������������Na2SO3���л�ԭ�ԣ���������ⷴӦ���������ᣬ������ؾ��������ԣ����������������������������������������Ȼ�����ˮ�ķֽ���Ӱ�죬�������������ʴ�Ϊ��a��
��5����

��Ӧת����֪����ΪAlԪ�أ���NΪAl2O3��ZΪAl��OH��3��MΪ�����ǽ���Ԫ�ص��Σ�ӦΪ��Σ���ӦX+YӦ�ǰ�����������ˮ��Һ�еķ�Ӧ��X��Һ��Y��Һ��Ӧ�����ӷ���ʽΪAl3++3NH3?H2O=Al��OH��3��+3NH4+����ҵұ�����õ���������ķ�������Ӧ����ʽΪ2Al2O3�����ڣ�
| ||
| ����ʯ |
�ʴ�Ϊ��Al3++3NH3?H2O=Al��OH��3��+3NH4+��2Al2O3�����ڣ�
| ||
| ����ʯ |
��6����Ӧ��=���յ�������-�ų���������������1mol N2����NH3�ķ�Ӧ��=946kJ+3��436 kJ-2��3��391kJ=-92kJ�������Ƿų��������ʴ�Ϊ���ų���92��

��ϰ��ϵ�д�
 �Ķ��쳵ϵ�д�
�Ķ��쳵ϵ�д�
�����Ŀ








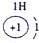
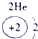
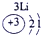
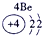
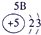
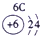
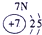
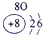
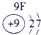
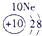
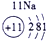





 ��ʾ����
��ʾ����